Matter is one of those words we hear very early in science, but it keeps getting more interesting the more you understand it. In simple terms, matter is the physical substance that makes up the things we can see, touch, measure, and study. It includes everything from a grain of sand to a mountain, from a glass of water to the air around you. Scientists also describe matter as anything that has mass and takes up space.
That sounds simple at first. But once you start looking closely, matter becomes a big idea that connects to atoms, molecules, states of matter, energy, and even the way the universe itself is built. In physics and chemistry, matter is not just a classroom definition. It is the starting point for understanding almost everything around us.
Table of Contents
Matter at a Glance
| Topic | Simple Meaning | Example |
|---|---|---|
| Matter | Anything with mass and volume | A chair, water, air |
| Atom | The basic building block of matter | A carbon atom, an oxygen atom |
| Molecule | Two or more atoms joined together | Water (H2O), carbon dioxide (CO2) |
| Mass | How much matter something contains | A textbook has more mass than a pencil |
| Weight | The force of gravity on an object | Your weight changes on the Moon |
| State of matter | The physical form matter takes | Solid, liquid, gas, plasma |
Matter is also important on a cosmic scale. Most normal matter is made of protons, neutrons, and electrons, and normal matter makes up less than 5% of the total universe.
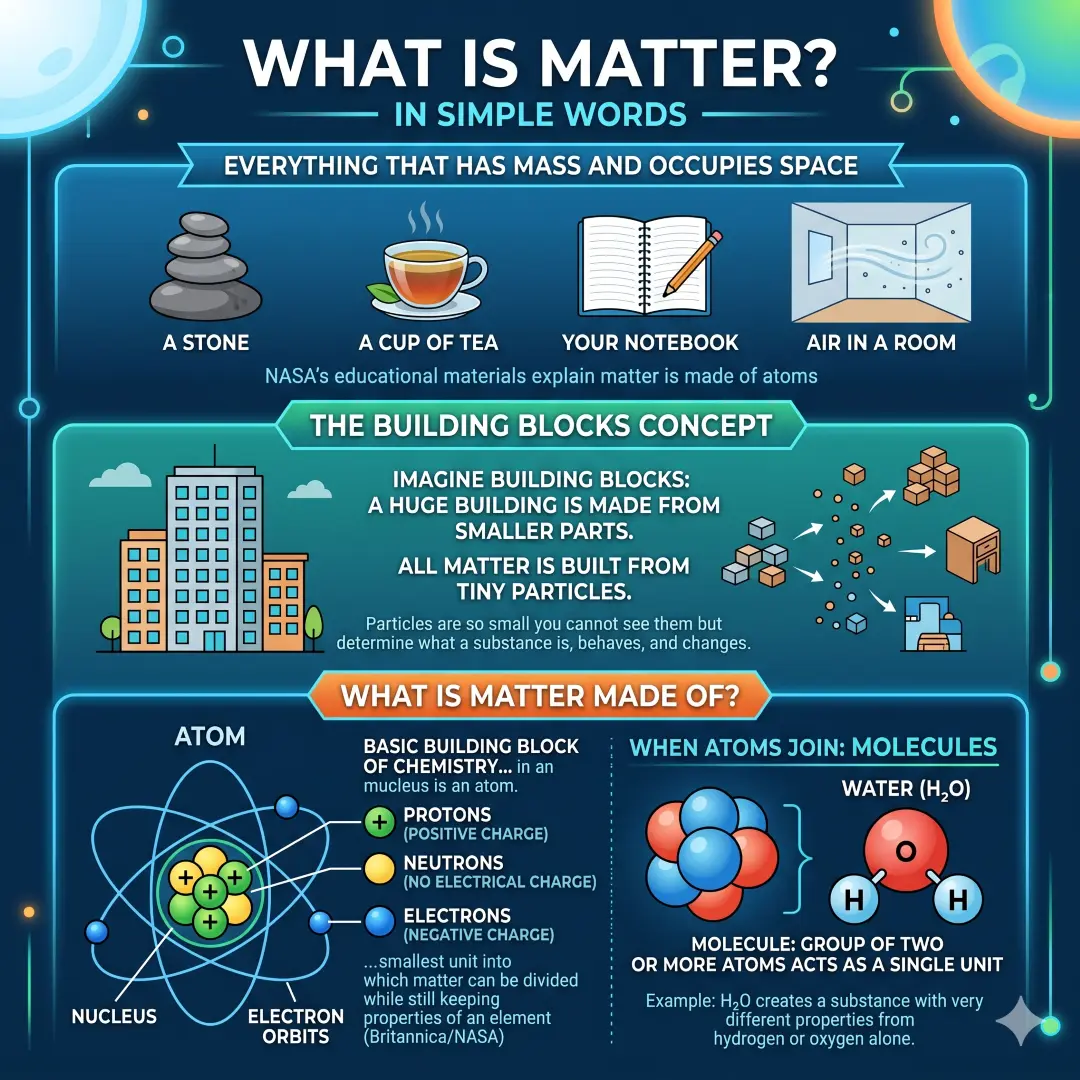
What Is Matter in Simple Words?
If you want the shortest, clearest answer, here it is. Matter is everything that has mass and occupies space. A stone is a matter. A cup of tea is a matter. Your notebook is a matter. The air in a room is matter too, even though you cannot see it. Matter is made of atoms, and those atoms combine to form the substances we know in daily life.
A useful way to think about matter is to imagine building blocks. A huge building is made from smaller parts. In the same way, all matter is built from tiny particles. These particles are so small that you cannot see them with your eyes, but they determine what a substance is, how it behaves, and how it changes.
What Is Matter Made Of?
A matter is made of atoms. An atom is the basic building block of chemistry and the smallest unit into which matter can be divided while still keeping the properties of an element. Atoms have a nucleus with protons and neutrons, and electrons around that nucleus.
When atoms join together, they form molecules. A molecule is a group of two or more atoms that acts as a single unit of a substance. For example, water is made from two hydrogen atoms and one oxygen atom, written as H2O. That small combination creates a substance with very different properties from hydrogen or oxygen alone.
The main building blocks of matter
- Protons carry a positive charge.
- Neutrons have no electrical charge.
- Electrons carry a negative charge.
- Atoms combine to form molecules and larger structures.
These tiny particles may sound abstract, but they are the reason a rock is hard, water flows, iron can rust, and salt tastes salty. Matter is not just about what something is made of. It is also about how those parts are arranged.
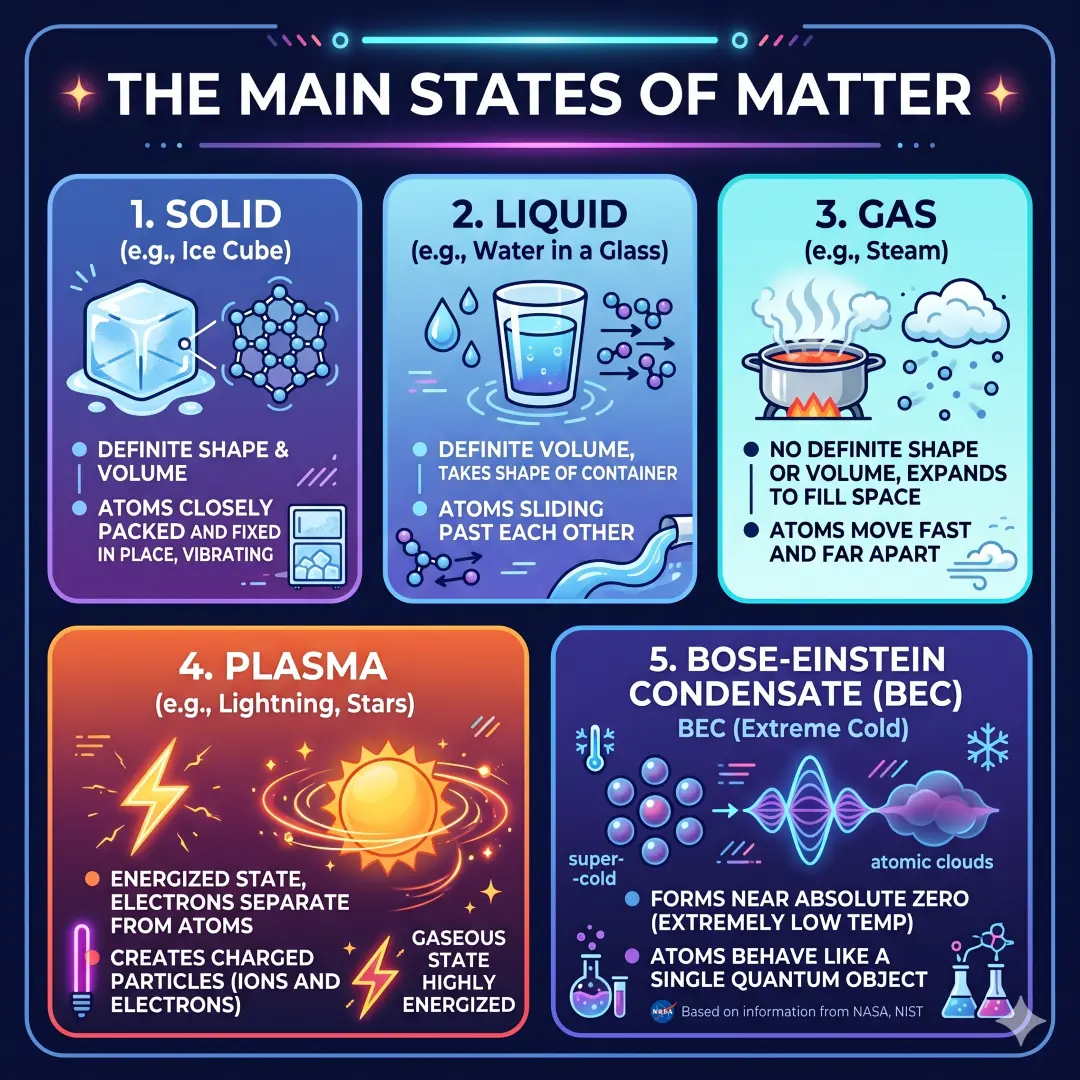
The Main States of Matter
On Earth, the most familiar states of matter are solid, liquid, and gas. Water in a glass is a liquid, ice in a freezer is a solid, and steam rising from hot water is a gas.
Scientists also recognize plasma as a fourth state of matter. Plasma forms when a gas gets so energized that its electrons separate from atoms, creating charged particles. Plasma is found in lightning, neon lights, and stars, including the Sun.
There is also a fifth state called a Bose-Einstein condensate (BEC), which appears at extremely low temperatures near absolute zero. When atoms are cooled enough, they can behave like a single quantum object.
Table: States of matter explained simply
| State | Shape | Volume | Particle behavior | Everyday example |
|---|---|---|---|---|
| Solid | Fixed | Fixed | Particles stay tightly packed and mostly vibrate in place | Ice, stone, wood |
| Liquid | Takes the shape of a container | Fixed | Particles move past one another | Water, milk, oil |
| Gas | No fixed shape | No fixed volume | Particles move freely and spread out | Air, steam |
| Plasma | No fixed shape | No fixed volume | Gas becomes ionized, with free charged particles | Lightning, the Sun, neon signs |
| Bose-Einstein condensate | Depends on conditions | Very small, quantum-like behavior | Particles act like one unified quantum state | Ultracold lab gases |
This table is a simplified view, but it captures the basic idea well. Matter can behave in very different ways depending on temperature, pressure, and the energy inside it.
Solids, Liquids, and Gases in Everyday Life
A solid keeps its shape because its particles are packed closely together. The particles still move a little, but not enough to let the solid flow easily. That is why a desk stays a desk, and a rock stays a rock. Solids are one of the familiar states where molecules are arranged in regular patterns and held in place by attractive forces.
A liquid has a fixed volume but does not have a fixed shape. It flows and takes the shape of its container. That is why water can sit in a bottle, a bowl, or a lake and still remain water.
A gas spreads out to fill the available space. Air is the easiest example. You do not notice it most of the time, but it surrounds you, fills rooms, and can be compressed. Gas is one of the fundamental states of matter with properties very different from solids and liquids.
Easy examples
- Ice is a solid because it keeps its shape.
- Milk is a liquid because it flows.
- Water vapor is a gas because it spreads out in air.
- Lightning is plasma because its particles are ionized and electrically charged.
What Are the Properties of Matter?
Matter has different properties, and these properties help us identify and describe substances. Some properties are easy to observe, like color and shape. Others need measuring tools, like density and mass. Matter is described by physical characteristics and by the behavior of its particles.
Table: Common properties of matter
| Property | Meaning | Example |
|---|---|---|
| Mass | Amount of matter in an object | A backpack full of books has more mass than an empty one |
| Volume | The space an object occupies | A balloon takes up more space when inflated |
| Density | How much mass is packed into a given volume | A metal coin is denser than a sponge |
| Shape | The form of an object | A ball, a cube, a sheet of paper |
| Color | Visual appearance | Red apple, blue pen |
| Texture | Surface feel | Smooth glass, rough sandpaper |
| Melting point | Temperature where a solid becomes liquid | Ice melts at 0°C at normal pressure |
| Boiling point | Temperature where a liquid becomes a gas | Water boils at 100°C at sea level |
These properties matter in daily life more than many people realize. When you cook, build, clean, or design something, you are working with the properties of matter all the time.
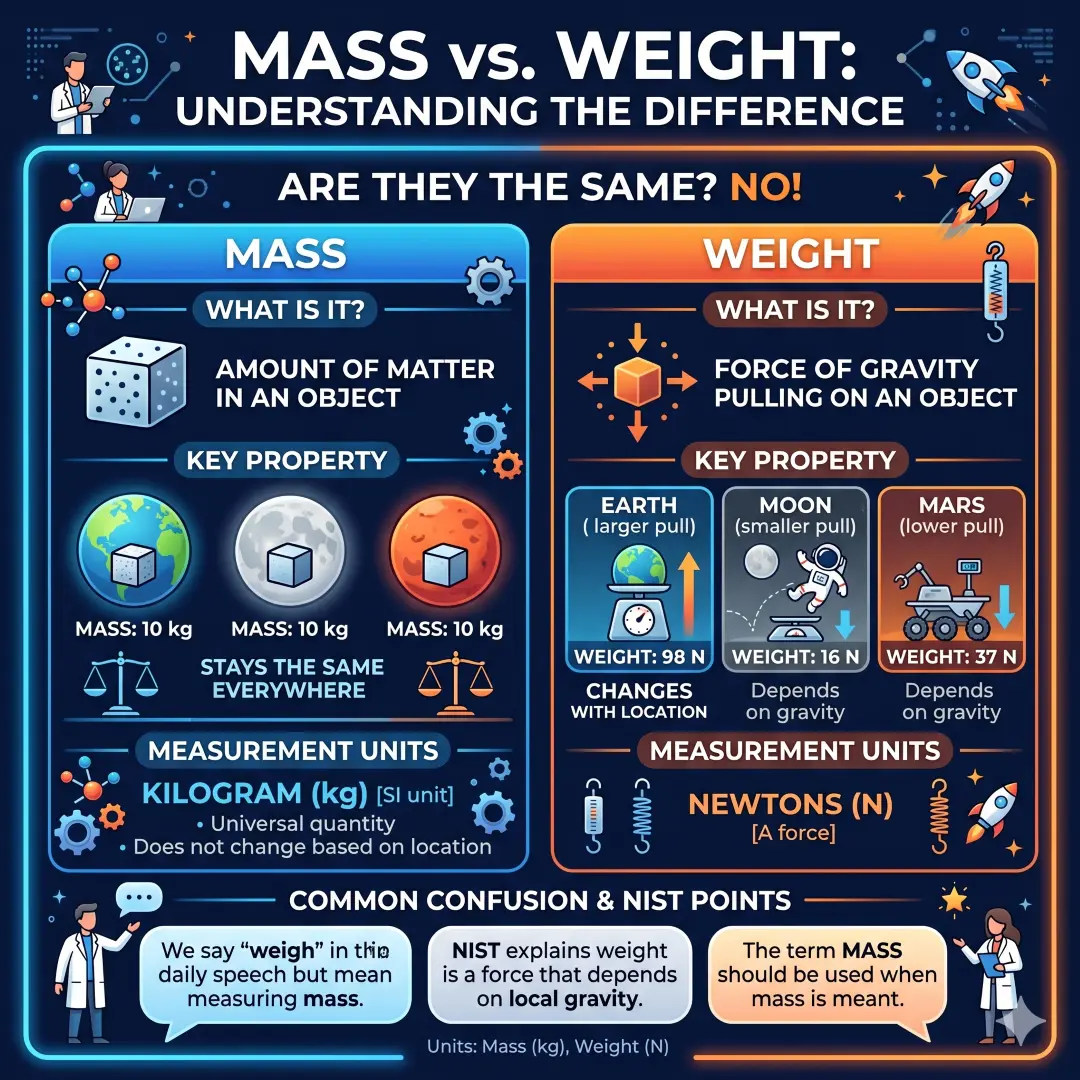
Mass and Weight Are Not the Same
This is one of the most common science mix-ups, so it deserves a clear explanation. Mass is the amount of matter in an object. Weight is the force of gravity pulling on that object. The SI unit of mass is the kilogram (kg), and weight is a force that depends on the local gravity field.
That means your mass stays the same whether you are on Earth, the Moon, or Mars. But your weight changes because gravity is different in each place. In everyday speech, people often say they “weigh” themselves when they really mean they are measuring mass. The term mass should be used when mass is meant.
Simple comparison
| Term | What it means | Changes with gravity? | SI unit |
|---|---|---|---|
| Mass | Amount of matter | No | kilogram (kg) |
| Weight | Force due to gravity | Yes | newton (N) |
This difference is important in science, engineering, space travel, and even shopping for groceries. A bag of rice has the same mass everywhere, but it does not “weigh” the same on every planet.
Matter and Energy
Matter and energy are closely connected, but they are not the same thing. Matter is a physical substance with mass and volume. Energy is the ability to do work or cause change.
This connection matters because matter can change form. Ice becomes water, water becomes steam, and under extreme conditions, gases become plasma. Matter can also be converted into powerful physical processes, which is why scientists study it so carefully. The relationship between matter and energy is one of the deepest ideas in physics.
Matter vs Energy table
| Aspect | Matter | Energy |
|---|---|---|
| What it is | Physical substance with mass and volume | Ability to do work or cause change |
| Can you touch it? | Often yes | Not in the same way |
| Examples | Rock, water, air | Light, heat, motion |
| Main role | Makes up physical objects | Makes processes happen |
A flame gives a good example. The wood and oxygen involved are matter. The heat and light released are forms of energy. Both are part of the same real-world event, but they play different roles.
Why Matter Is Important in Science
Matter is not just a topic in school science. It is the base layer for chemistry, physics, biology, astronomy, materials science, and engineering. Without matter, there would be no cells, no metals, no oceans, no rocks, and no living organisms as we know them.
Chemists study how atoms combine and rearrange to make new substances. Physicists study how matter behaves under different forces and conditions. Biologists study how living things use matter to grow, breathe, move, and reproduce. Engineers use matter to build stronger bridges, lighter aircraft, safer medicines, and better electronics.
Matter also shapes the natural world in obvious ways. Soil holds water and minerals. Rocks form landforms. Air supports weather. Water supports life. Every one of these systems depends on how matter is arranged and how it changes.
Matter in the Universe
On a cosmic scale, matter is everywhere, but not all matter is the same. Most normal matter is made up of atomic particles like protons, neutrons, and electrons, and normal matter can exist as gas, solid, liquid, or plasma. Normal matter makes up less than 5% of the total universe.
That is a striking fact. It means the stuff we touch every day is real and important, but it is only a tiny part of the whole cosmic picture. The universe contains much more than what we directly observe in ordinary life. Still, the matter we do see is what builds stars, planets, people, oceans, and cities.
Interesting facts about matter in space
- Stars are made mostly of hot plasma.
- Planets are made from a mix of solids, liquids, gases, and metals.
- Asteroids are large collections of matter shaped by gravity and collisions.
- Dust particles in space are tiny pieces of matter that help form larger bodies over time.
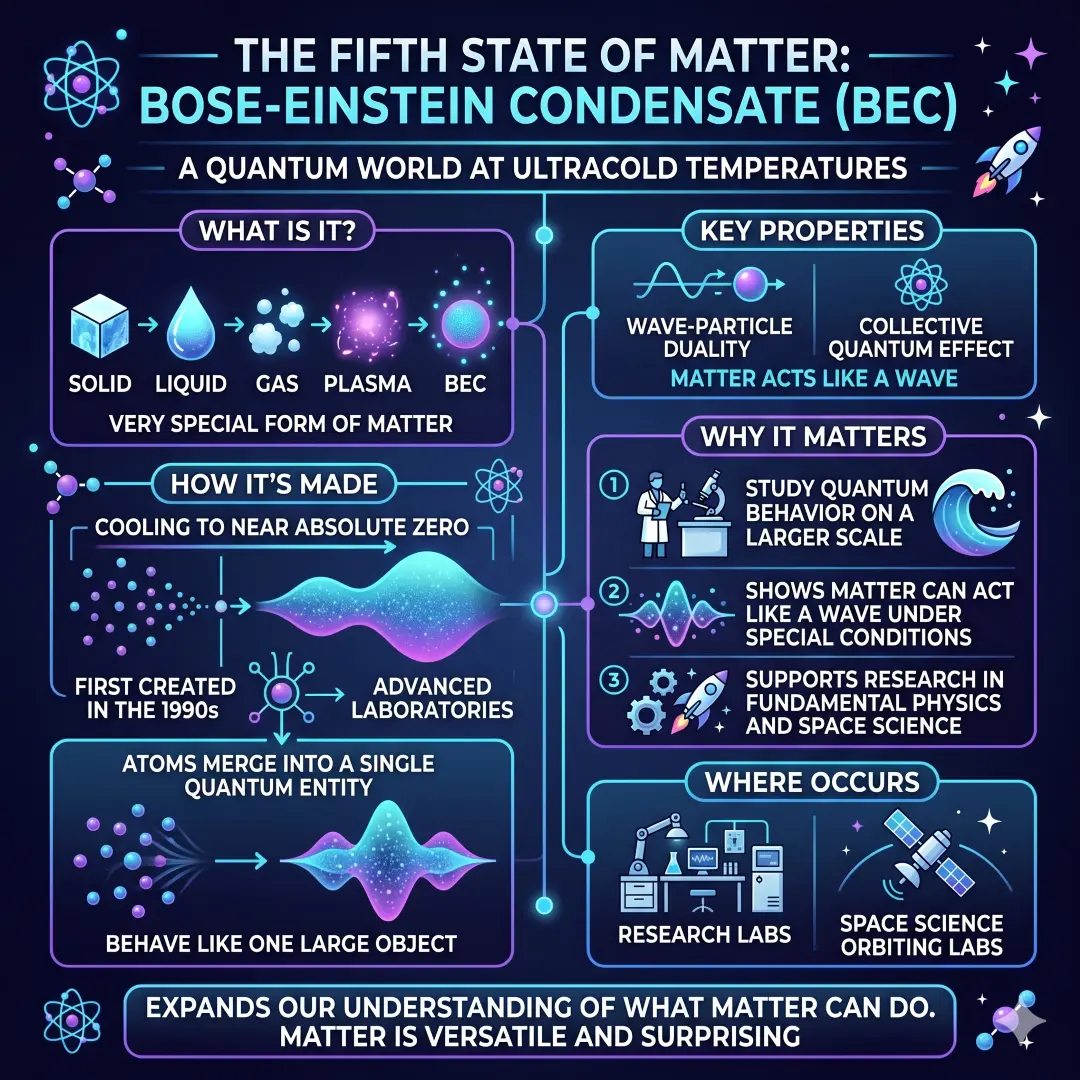
The Fifth State of Matter, Bose-Einstein Condensate
Most people learn about the first four states of matter, but modern physics goes further. Bose-Einstein condensate, often shortened to BEC, is a very special form of matter that appears at temperatures extremely close to absolute zero. When atoms are cooled to near absolute zero, they can merge into a single quantum entity and behave like one object.
This is not something you see in a kitchen or classroom. It happens in advanced laboratories. Scientists first created a BEC in the 1990s, and it became a major milestone in physics. Space laboratories have also produced this fifth state of matter in orbit.
Why BEC matters
- It helps scientists study quantum behavior on a larger scale.
- It shows how matter can act like a wave under special conditions.
- It supports research in fundamental physics and space science.
Even though BEC sounds remote from everyday life, it expands our understanding of what matter can do. That is what makes science exciting. Matter is not frozen into one simple behavior. Under the right conditions, it can surprise us.
Everyday Examples of Matter
Sometimes the best way to understand a concept is to look around your room. Almost everything nearby is a matter. Your phone, your desk, the chair you sit on, the glass of water beside you, the air in the room, and even your own body are all made of matter. Matter includes all visible physical substances and the atoms that compose them.
Practical examples
- Book → matter in solid form
- Tea → matter in liquid form
- Steam → matter in gas form
- Sunlight passing through a prism → energy interacting with matter
- Air in a balloon → matter that can be compressed and expanded
You can also think about matter in terms of human life. Food is matter, and your body uses it to build tissue and produce energy. Clothes are a matter. Medicine is a matter. Soil is matter. Water is matter. In a very real sense, daily life is a continuous relationship with matter.
How Matter Changes
Matter is not static. It changes all the time. Some changes are physical, like melting, freezing, evaporating, or condensing. Others are chemical, like rusting, burning, or cooking. The atoms are still involved, but they are arranged in new ways.
Table: Common changes in matter
| Change | What happens | Example |
|---|---|---|
| Melting | Solid becomes liquid | Ice turning into water |
| Freezing | Liquid becomes solid | Water turning into ice |
| Evaporation | Liquid becomes gas | A puddle drying up |
| Condensation | Gas becomes liquid | Water drops on a cold bottle |
| Sublimation | Solid becomes gas | Dry ice changing directly into gas |
| Ionization | Gas becomes plasma | Lightning or neon discharge |
These changes may look simple, but they reflect deeper particle behavior. When temperature changes, particles move differently. When pressure changes, they may pack together or spread out. That is how matter changes its form without losing its identity as matter.
A Clear Table of Matter Types and Everyday Samples
| Category | Main feature | Common samples | Where you see it |
|---|---|---|---|
| Natural matter | Found in nature without human assembly | Rocks, water, air, wood | Forests, rivers, mountains |
| Living matter | Part of living organisms | Plants, animals, bacteria | Gardens, bodies, ecosystems |
| Man-made matter | Shaped or made by people | Plastic, glass, steel, paper | Homes, tools, devices |
| Ordinary matter | What we can directly observe | Everything made of atoms and particles | Daily surroundings |
This table makes one thing clear. Matter is not limited to one kind of thing. It includes natural, living, and human-made substances. The same basic physical rule applies across all of them. If it has mass and occupies space, it belongs to the world of matter.
Common Misconceptions About Matter
A lot of confusion around matter comes from everyday language. People often say that something “does not matter” or that “it disappeared,” but science works differently. If something truly disappears as a substance, it usually changes form or becomes harder to detect, rather than simply vanishing from existence. Matter can be transformed, rearranged, or converted into different states, but its study always begins with measurement and observation.
Another common mistake is confusing matter with stuff in a loose sense. In science, matter has a precise meaning. It is not just any abstract thing. It is a physical substance with mass and volume. That definition helps scientists compare objects, test materials, and explain natural processes with consistency.
Mistakes to avoid
- Thinking mass and weight are the same thing
- Assuming only solids count as matter
- Forgetting that air is matter
- Believing matter cannot change form
- Mixing up matter with energy
Why Matter Matters in Real Life
Matter is one of those ideas that sounds abstract until you notice how practical it is. Builders need to know how matter behaves when they use concrete, steel, and glass. Doctors need to understand matter when they study blood, medicine, and tissues. Farmers need to understand soil and water. Chefs need to understand heat, moisture, and ingredients. Designers need to understand weight, texture, and durability.
Even simple decisions depend on matter. A plastic bottle is lighter than a glass bottle. A sponge absorbs liquid because of its structure. A metal pan heats differently from a wooden spoon. A balloon expands because gas particles move freely. All of these everyday observations come from the same basic science of matter.
Matter also shapes how we think about the world. Once you understand that everything physical is made of tiny particles, ordinary things become more interesting. A drop of water is no longer just a drop of water. It is a collection of molecules, moving, interacting, and obeying natural laws. That perspective makes the world feel both simpler and richer.
Quick Reference Guide
| Question | Simple answer |
|---|---|
| What is matter? | Anything that has mass and takes up space |
| What is matter made of? | Atoms and molecules |
| What are the main states of matter? | Solid, liquid, gas, plasma |
| Is air matter? | Yes, air has mass and volume |
| Is light matter? | No, light is energy |
| Is weight the same as mass? | No, weight depends on gravity |
| What is the fifth state of matter? | Bose-Einstein condensate |
This kind of quick summary can be useful for students, teachers, and anyone who wants the core idea in one place. The deeper explanation still matters, but the basic definition is where everything begins.
Final Thoughts
So, what is matter? It is the physical substance that makes up the universe we can observe. It includes everything with mass and volume, from tiny atoms to massive planets. It appears in familiar forms like solids, liquids, and gases, but it can also become plasma or, in special lab conditions, Bose-Einstein condensate. Matter is not just a school topic. It is the foundation of science, nature, technology, and daily life.
And maybe that is the nicest thing about this topic. Matter is everywhere, but it never really becomes boring. The more you learn about it, the more the world makes sense. A cup, a cloud, a cell, a star, and even the air in your room are all part of one big physical story. That story starts with matter.
Article References and Sources
- Matter: Definition and Overview
- Atom: Basic Building Block of Matter
- Molecule: Structure and Examples
- Gas State of Matter: Explanation
- Understanding Matter (Student Resource)
- NASA: Building Blocks of the Universe
- NASA: States of Matter and Plasma
- NASA: Atoms and Matter Basics
- NASA Glenn Research Center: States of Matter
- NIST: SI Units and Mass Explanation
- NIST: Bose-Einstein Condensate Research
- NASA: What Is the Universe?
Frequently Asked Questions
FAQ 1: What is matter in simple words?
Matter is anything that has mass and takes up space. That is the easiest way to understand it, and it covers almost everything around us. A desk is matter. Water is matter. Air is matter. Your body is matter. Even though some things are easy to see and touch, and others are invisible, they can still be matter as long as they have mass and volume.
The idea becomes much clearer when you think about daily life. A stone placed on the ground is matter because it occupies space and has weight because of gravity. A glass of juice is matter because it fills a container and has substance. The air in a balloon is matter too, even though you cannot hold it in your hand the same way you hold a rock. The balloon expands because the air inside it takes up space.
Matter is not just about what you can see. It also includes things that are too small to see, like atoms and molecules. These tiny building blocks join together to form everything physical in the world. So when people ask what matter is, the real answer is simple and deep at the same time. It is the physical stuff that makes up the universe we live in.
And that is why matter matters so much in science. It is the starting point for understanding nature, objects, living things, and even the stars in space. Once you understand matter, you start to understand how the physical world works.
FAQ 2: What are the main states of matter?
The main states of matter are solid, liquid, gas, and plasma. In some scientific settings, people also talk about a fifth state called Bose-Einstein condensate. Each state has different behavior, even though all of them are still matter.
A solid has a fixed shape and a fixed volume. Its particles are packed closely together, so it does not flow easily. A chair, a rock, and a book are all solids. A liquid has a fixed volume but no fixed shape. It takes the shape of its container. Water, milk, and oil are liquids. A gas has no fixed shape and no fixed volume. It spreads out to fill the space around it. Air is the most familiar example. Plasma is similar to gas, but it is made of charged particles. It appears in lightning, the Sun, and neon signs.
These states are not separate kinds of matter. They are different ways matter behaves depending on temperature and pressure. For example, ice can melt into water, and water can turn into steam. That means the same substance can move from one state to another without stopping being matter.
The fifth state, Bose-Einstein condensate, happens only under extreme cold conditions in laboratory settings. It is a fascinating state where particles behave almost like one single unit. Most people will never see it in daily life, but it is very important in advanced physics.
So when you think about states of matter, remember that they are really about how particles behave. Matter can be hard, flowing, invisible, or highly energized. The form changes, but the basic idea remains the same.
FAQ 3: What is matter made of?
Matter is made of extremely small particles called atoms. These are the basic building blocks of all ordinary matter. Atoms are so tiny that you cannot see them with your eyes, but everything physical is built from them. A single atom can be part of an element, and many atoms can join together to make larger structures.
Inside an atom, there are three main particles: protons, neutrons, and electrons. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge. Protons and neutrons stay in the center of the atom, which is called the nucleus, while electrons move around the nucleus. This structure helps explain why matter behaves the way it does.
When atoms connect with each other, they form molecules. A molecule is a group of atoms bonded together. For example, water is made of two hydrogen atoms and one oxygen atom. That small combination creates a substance with very different properties from the individual elements. This is one of the most interesting things about matter. Tiny particles can join in different ways and create entirely new materials.
Matter can also be made of larger structures built from atoms and molecules. Wood, metal, plastic, and living tissue all come from atomic and molecular arrangements. So matter is not one single thing. It is a huge family of physical substances made from tiny building blocks working together in different patterns.
And that is what makes the study of matter so rich. Once you understand atoms, you begin to understand how all physical things are formed.
FAQ 4: What is the difference between mass and weight?
Mass and weight are not the same thing, even though people often use them as if they are. Mass is the amount of matter in an object. Weight is the force of gravity pulling on that object. That is the key difference.
A bag of rice has the same mass whether it is on Earth, the Moon, or Mars. But its weight changes because gravity is different in those places. On Earth, gravity is stronger than on the Moon, so the same object would weigh less there. This is why astronauts feel lighter in space. Their mass stays the same, but the force of gravity acting on them changes.
Mass is measured in kilograms (kg), which is the standard scientific unit. Weight is measured in newtons (N) because it is a force. That difference is important in science, engineering, and daily life. A doctor, a scientist, and a shopkeeper may all use measurements, but they may be talking about mass, weight, or both depending on the situation.
A simple way to remember it is this. Mass tells you how much matter is inside something. Weight tells you how strongly gravity pulls on it. If gravity disappeared, mass would still exist, but weight would not in the same way.
This distinction helps people understand everything from bathroom scales to space travel. It is a small idea with a big impact.
FAQ 5: Is air matter even though we cannot see it?
Yes, air is matter. This surprises some people at first because air is invisible, but invisibility does not mean something is not matter. Air has mass and it takes up space, which means it fits the scientific definition of matter perfectly.
You can prove this in simple ways. A balloon filled with air is heavier than an empty balloon. That happens because the air adds mass. A tire becomes firm when filled with air because the gas inside occupies space and pushes outward. Wind can move leaves, flags, and even buildings in strong storms because air is real physical substance, not empty nothingness.
Air is mostly made of gases like nitrogen and oxygen, along with smaller amounts of other gases. These gas particles are moving all the time. They spread out and fill containers. That is one of the reasons air behaves differently from solids and liquids. You do not see the particles easily, but they are still there.
This is a good reminder that matter is not only what looks solid or heavy. Some matter is light, spread out, and hard to notice. Air is one of the best examples. It is everywhere, supports life, and constantly interacts with our bodies, weather, and environment.
So yes, air is definitely matter. It is just matter in a gas form.
FAQ 6: How does matter change from one state to another?
Matter changes from one state to another when its particles gain or lose energy, usually because of changes in temperature or pressure. This is why ice can melt into water, and water can turn into steam. The substance is still matter, but its form changes.
When a solid gets warmer, its particles move faster. If they get enough energy, the solid can melt into a liquid. When a liquid is heated, its particles move even faster and can escape into the air as a gas through evaporation or boiling. When a gas cools down, it can turn back into a liquid through condensation. If a liquid cools even more, it can become a solid through freezing.
These changes happen all the time in nature. Rain forms when water vapor condenses in the sky. Ice forms when water freezes in cold weather. Puddles disappear when the water evaporates into the air. Dry ice changes directly from solid to gas through sublimation. Each of these changes is a shift in state, not a disappearance of matter.
The important thing to remember is that matter does not stop being matter just because it changes form. The particles are still there. They are simply moving differently and arranged differently. This is one of the most basic and useful ideas in science.
And once you notice it, the world starts to look more connected. Cooking, weather, climate, and even industrial processes all depend on how matter changes state.
FAQ 7: What are the properties of matter?
The properties of matter are the qualities that help us describe and identify substances. Some properties are easy to see, while others need special tools to measure. These properties tell us what a substance is like and how it behaves.
Common properties include mass, volume, density, shape, texture, color, melting point, and boiling point. Mass tells us how much matter is in something. Volume tells us how much space it occupies. Density tells us how tightly that matter is packed together. A metal coin is denser than a sponge because more matter is packed into a smaller space.
Other properties help us understand how matter behaves under different conditions. The melting point tells us when a solid becomes a liquid. The boiling point tells us when a liquid becomes a gas. Texture tells us whether a surface feels rough, smooth, soft, or hard. Color helps us identify materials visually.
These properties matter in real life more than people usually notice. Builders care about strength and density. Chefs care about melting and boiling points. Doctors care about the properties of blood, tissue, and medicine. Scientists use these properties to identify unknown materials and study how substances change.
So when we talk about matter, we are not just talking about “stuff.” We are talking about physical substances with specific traits that can be observed, measured, and compared.
FAQ 8: What is the difference between matter and energy?
Matter and energy are related, but they are not the same thing. Matter is physical substance that has mass and occupies space. Energy is the ability to do work or cause change. That means matter is the thing, while energy is what makes things happen.
A good example is a burning candle. The wax and wick are matter. The flame gives off light and heat, which are forms of energy. The candle uses matter as fuel, and energy is released through the process of burning. Both are involved, but they play different roles.
Another example is food. Food is matter. When your body digests it, the chemical energy stored in the food becomes useful energy for movement, growth, and life processes. That is why humans need both matter and energy. Matter builds the body, and energy powers its functions.
Light is often confused with matter, but light is energy, not matter. It does not have rest mass in the same way physical objects do, and it behaves differently from solid substances or gases. This is why scientists carefully separate the two ideas.
Still, matter and energy interact constantly. In the physical world, they are part of the same big picture. You cannot fully understand one without at least thinking about the other. That is why both are central ideas in science.
FAQ 9: Why is matter important in everyday life?
Matter is important in everyday life because everything physical around us is made of matter. Your house, your clothes, your food, your phone, your chair, your school bag, and your body are all matter. Life as we know it depends on matter in countless ways.
Think about a normal day. You wake up on a bed made of matter. You brush your teeth with a toothbrush made of matter. You drink water, which is matter. You eat food, which is matter. You travel in a vehicle built from metal, glass, rubber, and plastic, all of which are forms of matter. Even the air you breathe is matter.
Matter is also important in work and technology. Builders use matter to create houses and roads. Engineers use matter to design machines and bridges. Doctors rely on matter to treat the human body. Farmers depend on soil and water. Teachers use paper, pens, whiteboards, and devices, all made from matter in different forms.
What makes matter especially useful is that it can be shaped, combined, heated, cooled, stretched, compressed, and transformed. That means humans can use matter to create almost everything in modern life. Without matter, there would be no tools, no cities, and no living organisms in the way we understand them.
So matter is not just a science word. It is the foundation of ordinary life.
FAQ 10: Why do scientists study matter so carefully?
Scientists study matter carefully because it is the foundation of the physical universe. If you want to understand how the world works, you have to understand what things are made of and how they behave. Matter is the starting point for that study.
Chemists study how atoms combine to form new substances. Physicists study how matter responds to forces, heat, motion, and energy. Biologists study how living matter works inside cells, tissues, and organisms. Astronomers study matter in stars, planets, dust clouds, and galaxies. Each field looks at matter in a different way, but the core idea is the same.
Scientists also study matter to solve practical problems. They want to create stronger materials, cleaner energy sources, better medicines, safer buildings, and more efficient technology. To do that, they need to understand properties like density, conductivity, melting point, pressure, and atomic structure.
Matter is also important because it helps answer deep questions. What is the universe made of? How do stars form? How does life grow? Why do some materials conduct electricity while others do not? These questions all lead back to matter and its behavior.
And that is why the study of matter never gets old. It connects everyday objects to the biggest questions in science. It helps us understand the small things and the huge things at the same time.