Thermal energy is one of the most familiar forms of energy in daily life. You feel it when sunlight warms your skin, when a cup of tea cools down on the table, when a stove heats a pan, and when insulation helps keep a room comfortable. In simple terms, thermal energy is the energy linked to the motion of atoms and molecules inside matter. The faster and more randomly those particles move, the more thermal energy a substance has in a practical sense.
People often mix up thermal energy, heat, and temperature, but they are not the same thing. Temperature tells us about the average energy of particles in a substance, while heat is energy that moves from a warmer object to a cooler one because of a temperature difference. Thermal energy is the broader everyday idea that helps explain both of those concepts.
This article gives a full, human-friendly look at thermal energy, how it works, how it moves, where it appears in nature, how it powers modern life, and why it matters so much in homes, cities, industry, and the environment. It also covers storage, efficiency, and real-world examples in a way that is easy to read but still scientifically solid.
Table of Contents
What Thermal Energy Means
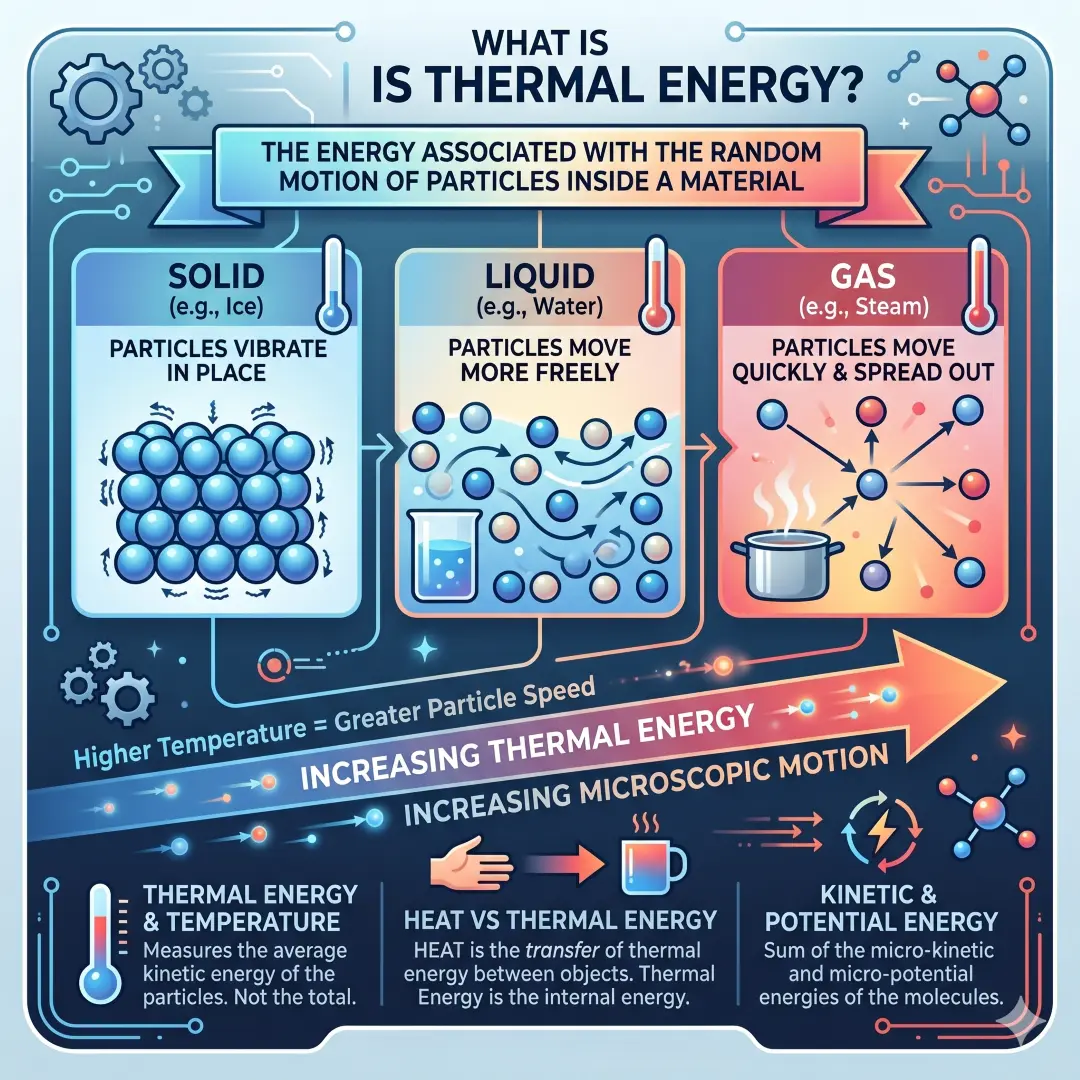
Thermal energy is the energy associated with the random motion of particles inside a material. In a solid, the particles vibrate in place. In a liquid, they move more freely. In a gas, they move quickly and spread out even more. The more energetic that microscopic motion becomes, the more thermal energy the substance contains.
A helpful way to think about it is this. A hot object usually has particles moving more rapidly than a cold object. That is why the hot object can transfer energy to the cooler one. This transfer is what we call heat. So thermal energy is tied to what is happening inside the material, while heat is the movement of energy between materials.
Thermal energy is not just about “feeling hot.” It also explains melting, boiling, cooling, insulation, weather patterns, cooking, power generation, and even how the Earth stores and releases energy. In other words, thermal energy is a quiet force behind many ordinary things we take for granted.
Thermal Energy, Heat, Temperature, and Internal Energy
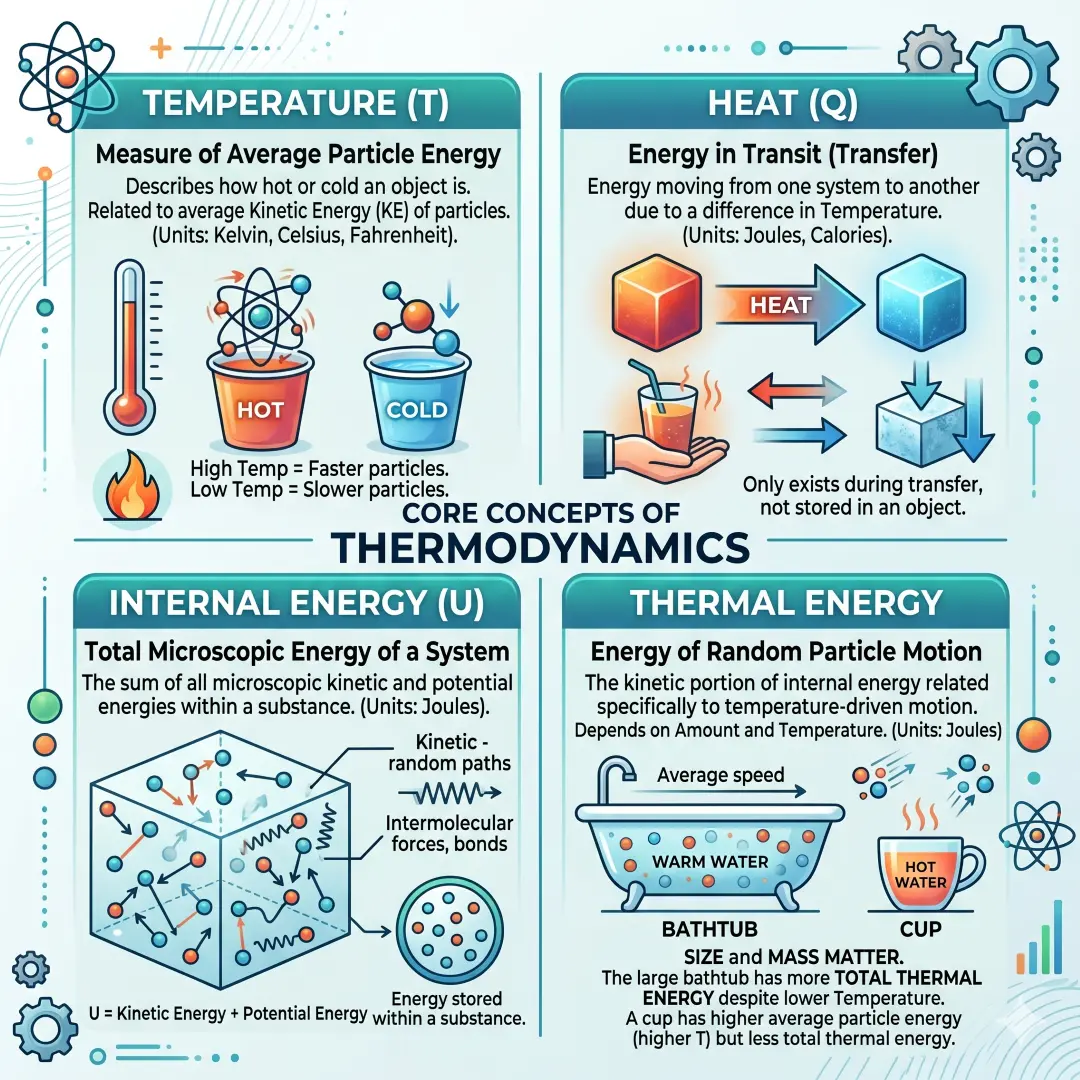
These four ideas are closely related, so it helps to separate them clearly.
Temperature is a measure of the average energy of particles in a system. Heat is energy in transit, moving because of a temperature difference. Internal energy is the total microscopic energy contained in a system, including particle motion and other contributions. Thermal energy is often used in everyday science writing as the energy tied to particle motion inside matter, especially when discussing heating and cooling.
That means a cup of water can have a high temperature but still contain less total thermal energy than a large bathtub of warm water. Size matters. Mass matters. Substance matters. A tiny flame can be very hot, but it does not necessarily store as much thermal energy as a large warm object.
Here is a simple table that makes the differences easier to see.
| Term | Simple meaning | How it behaves | Easy example | Why it matters |
|---|---|---|---|---|
| Thermal energy | Energy is linked to the motion of particles inside matter | Usually rises when particles move faster | Warm soup has more thermal energy than cold soup | Helps explain heating, cooling, and energy storage |
| Heat | Energy is transferred from a warmer place to a cooler place | Exists only during transfer | A hot mug warming your hands | Important for cooking, weather, insulation, and engines |
| Temperature | A measure of the average energy of particles | Tells how hot or cold something is | Water at 80°C is hotter than water at 20°C | Used in science, medicine, weather, and engineering |
| Internal energy | Total microscopic energy inside a system | Includes particle motion and other microscopic contributions | A gas in a sealed container has internal energy | Central idea in thermodynamics and energy balance |
How Thermal Energy Moves
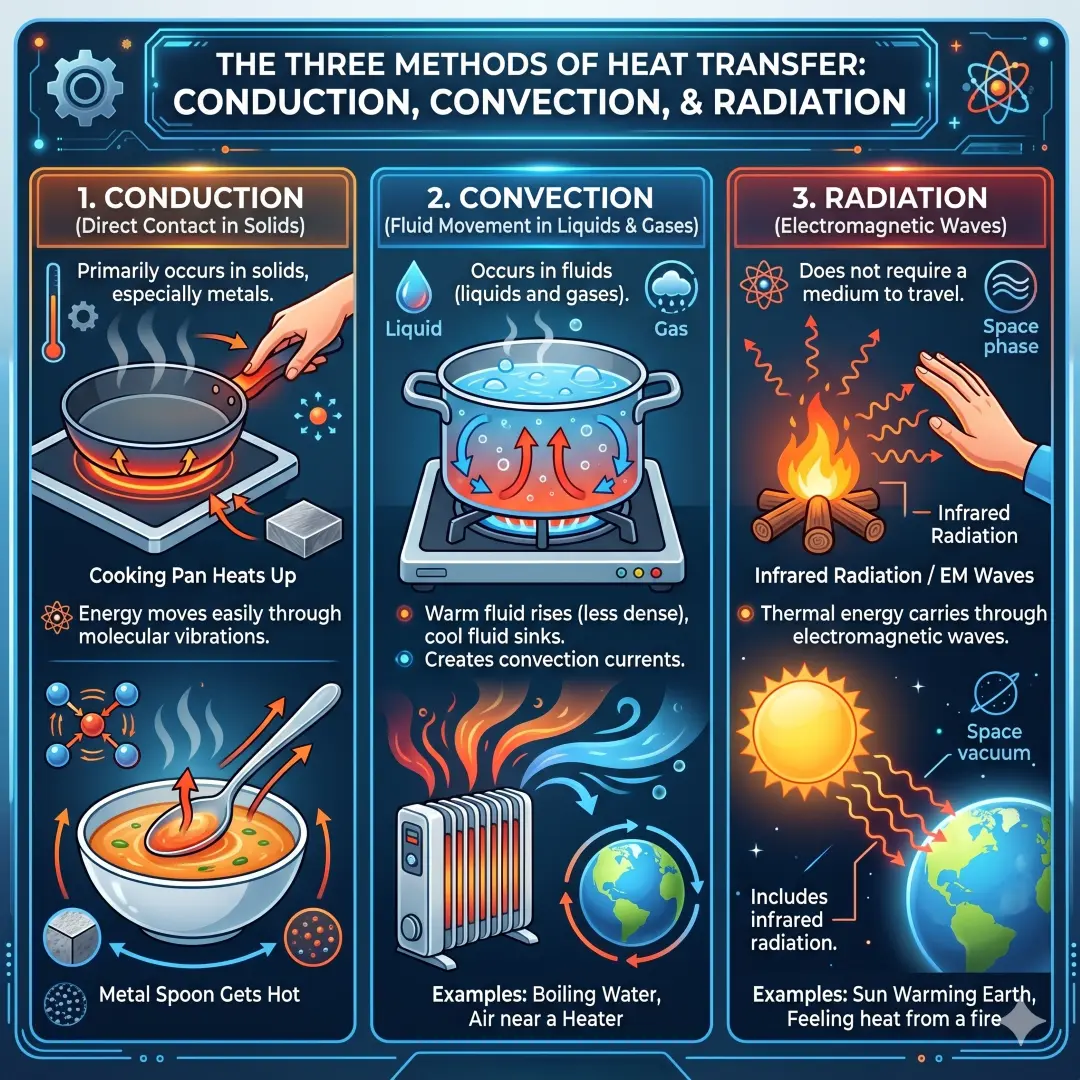
Thermal energy moves in three main ways, and these three mechanisms shape almost everything from home comfort to climate science.
The first is conduction, which is heat flow through direct contact. The second is convection, which happens when fluids such as air or water move and carry energy with them. The third is radiation, which transfers energy by electromagnetic waves and does not need direct contact or even a material medium.
That is why a metal spoon gets hot in a pan, why boiling water circulates, and why sunlight can warm the Earth from space. These are not separate “kinds of heat” so much as different routes for thermal energy transfer.
Table: The Three Main Modes of Heat Transfer
| Mode | How it works | Where it happens most | Common example | Key idea |
|---|---|---|---|---|
| Conduction | Energy moves through direct contact between particles | Solids, especially metals | A spoon in hot coffee gets warm | Faster in materials that conduct well |
| Convection | Warm fluid rises and cooler fluid sinks, creating circulation | Liquids and gases | Warm air rising in a room | Strongly linked to fluid motion |
| Radiation | Energy travels as electromagnetic waves | Works through vacuum and matter | Sunlight warming skin | Does not need direct contact |
A closer look at conduction
Conduction is especially important in solids. Metals are often excellent conductors because energy moves easily through their structure. That is why a cooking pan heats up quickly and why a metal spoon left in soup does not stay cool for long. In homes, conduction is also one of the major ways unwanted heat enters during hot weather or escapes during cold weather.
A closer look at convection
Convection happens in fluids, which means liquids and gases. When part of a fluid warms up, it becomes less dense and rises. Cooler fluid sinks to replace it, creating a current. This is why warm air near a heater moves upward and why boiling water circulates inside a pot.
A closer look at radiation
Radiation carries thermal energy through electromagnetic waves, including infrared radiation. This is why you can feel heat from a fire or stovetop without touching it. It is also why the Sun can heat Earth across the vacuum of space.
Why Temperature Does Not Tell the Whole Story
A common mistake is assuming that a higher temperature always means more thermal energy overall. That is not true. A small object can be much hotter than a large one but still contain less total energy. The total amount of thermal energy depends on the mass, the material, and the temperature change involved.
This is one reason a large pot of warm water can feel like it holds more heat than a small metal bolt that is glowing hot. The bolt may be at a higher temperature, but the pot may store far more thermal energy because it contains far more matter.
Table: Why Temperature and Thermal Energy Are Not the Same
| Situation | Temperature | Thermal energy | What this teaches us |
|---|---|---|---|
| Small metal object, very hot | Very high | May be low overall | Temperature alone does not show total energy |
| Large container of warm water | Moderate | Often very high overall | Mass can matter more than peak temperature |
| Room air and a heated wall | Similar average temperature | Different energy storage and transfer behavior | Materials store and move energy differently |
Thermal Energy in Daily Life
You do not need a lab to see thermal energy in action. You meet it every day.
A kitchen stove converts energy into thermal energy that cooks food. A thermos flask slows the escape of thermal energy. A blanket reduces heat loss from your body. A car radiator helps remove unwanted thermal energy from the engine. A smart building uses insulation and window design to control heat flow.
And it is not just about comfort. Thermal energy affects how food changes texture, how quickly water boils, how engines run, and how buildings stay efficient. That makes it one of the most practical concepts in all of science.
Examples you already know
- Boiling water needs thermal energy to raise its temperature and change state.
- Ice melting absorbs thermal energy before the temperature rises much further.
- A hot cup of tea cools because thermal energy moves into the room.
- A metal spoon in soup warms through conduction.
- Sunlight through a window delivers thermal energy by radiation.
How Thermal Energy Changes with Matter
Different materials do not respond to thermal energy in the same way. Some warm up quickly. Some stores heat well. Some conduct heat very efficiently. Some act as good insulators.
This is why water is such a useful thermal medium. It is widely used in cooling systems, heating systems, and thermal storage because it can store and move energy effectively. In energy storage systems, the amount of stored energy depends on properties such as density, volume, specific heat, and temperature change.
Table: Materials and Thermal Behavior
| Material type | Thermal behavior | Typical use | Plain-language explanation |
|---|---|---|---|
| Metals | Usually good conductors | Pots, pans, heat exchangers | They pass energy through quickly |
| Water | High practical heat-storage value | Heating, cooling, storage tanks | It can absorb and release a lot of thermal energy |
| Air | Poor conductor, easy to move by convection | Insulation, ventilation, weather | Air pockets slow heat flow when trapped in insulation |
| Insulation materials | Designed to resist heat flow | Walls, roofs, attics | They slow conduction, convection, and sometimes radiation |
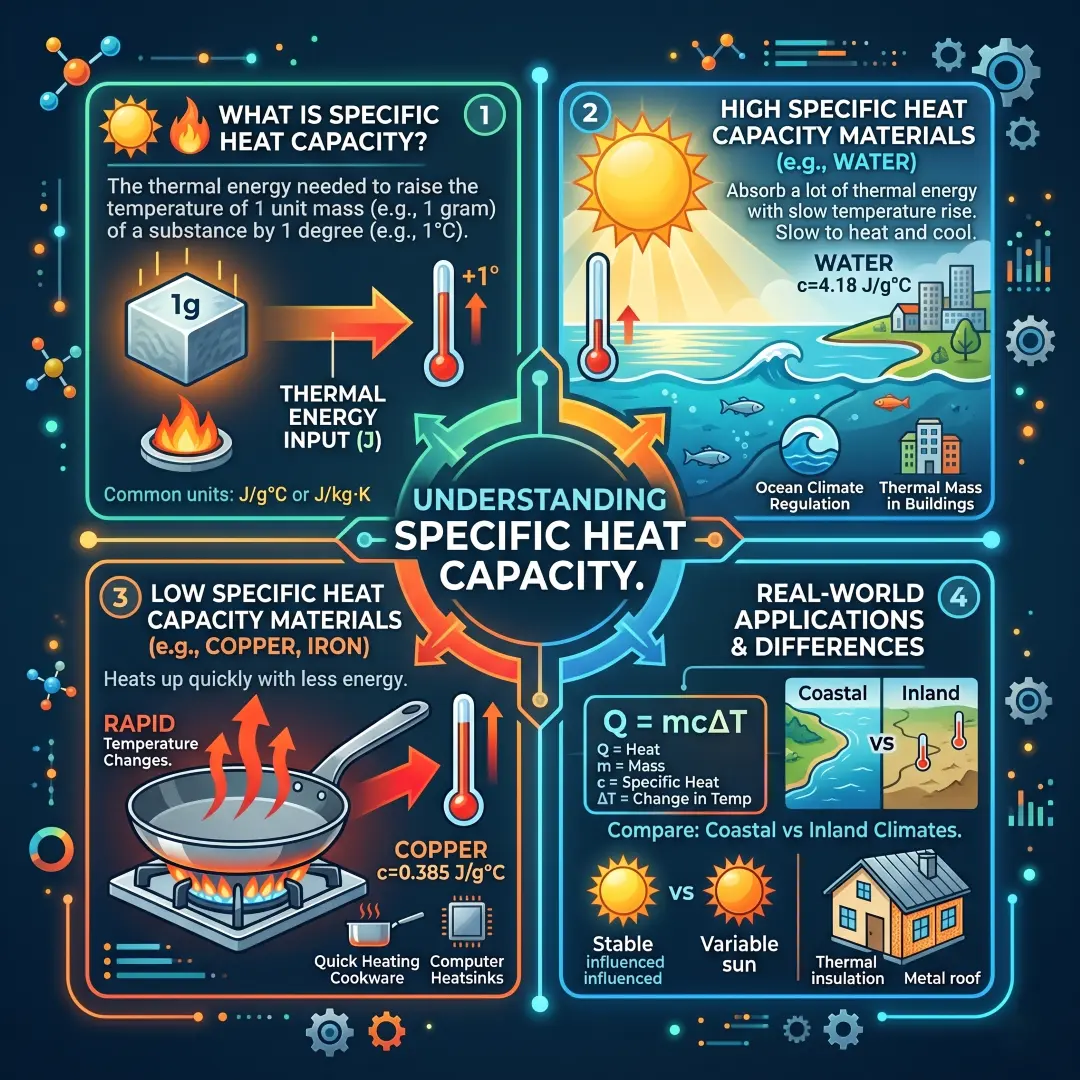
Specific Heat Capacity and Why It Matters
Specific heat capacity tells us how much thermal energy is needed to raise the temperature of a unit mass of a substance by one degree. Materials with high specific heat can absorb a lot of thermal energy before their temperature rises very much.
This is one reason water plays such a big role in climate, weather, and engineering. It changes temperature more slowly than many other substances, so it can absorb and release large amounts of heat without dramatic temperature swings. That makes it useful in cooling systems, storage systems, and nature itself.
Here is a practical way to think about it. If two objects receive the same amount of energy, the one with lower specific heat usually warms up more quickly. That is why different materials feel different under the same sunlight, and why some surfaces get hot much faster than others.
Phase Changes and Latent Heat
Thermal energy does not always show up as a simple temperature rise. Sometimes it goes into changing the state of matter, such as melting, freezing, boiling, or condensing. During these changes, energy can be absorbed or released while the temperature stays nearly steady for a time.
This hidden energy is often called latent heat. It matters in cooking, weather, ice storage, steam systems, and building comfort. A familiar example is ice. Before ice becomes liquid water, it absorbs energy that goes into the change of phase rather than a big temperature rise.
Table: Thermal Energy During Phase Changes
| Process | What thermal energy does | What happens to the temperature | Everyday example |
|---|---|---|---|
| Melting | Energy is absorbed | The temperature may stay nearly steady during the change | Ice cubes in a drink |
| Freezing | Energy is released | The temperature stays near the freezing point during the change | Water turning to ice in a freezer |
| Boiling | Energy is absorbed | Temperature may stay near the boiling point while the liquid becomes vapor | Water boiling for tea |
| Condensation | Energy is released | Vapor loses energy and becomes liquid | Water droplets forming on a cold glass |
The Joule and the Measurement of Thermal Energy
In science, the standard unit for energy, work, and quantity of heat is the joule. That means thermal energy is measured in the same unit as other forms of energy. This helps scientists and engineers compare heat with motion, electricity, and mechanical work on the same scale.
The historical shift toward using the joule for heat helped make energy measurements more consistent and easier to compare. Today, that same unit is used across physics, engineering, and thermodynamics.
Table: Common Thermal Energy Measurement Ideas
| Quantity | What it describes | Standard idea | Why it is useful |
|---|---|---|---|
| Joule | The SI unit of energy and heat | Used for scientific measurement | Keeps energy comparisons consistent |
| Specific heat capacity | Energy needed to raise the temperature of a unit mass by 1 degree | Depends on the substance | Helps predict heating and cooling behavior |
| Temperature change | Difference between start and end temperature | Measured in kelvins or degrees Celsius | Tells how much a body warms or cools |
Thermal Energy in Homes and Buildings
Buildings are full of heat flow. Heat enters through roofs, walls, windows, and doors. It leaves through the same paths. That is why insulation, window design, air sealing, and reflective materials matter so much. The goal is not to stop thermal energy completely. The goal is to control it.
Insulation works by resisting heat flow. It slows conduction, reduces convection, and can also reduce radiant heat gain depending on the system. That is why proper insulation lowers heating and cooling costs and improves comfort.
In modern buildings, thermal control is not just a comfort feature. It is an efficiency strategy. Less unwanted heat loss in winter means lower energy use. Less unwanted heat gain in summer means less air-conditioning demand.
Thermal Energy in Weather and Climate
The weather is deeply connected to thermal energy. The Sun heats Earth unevenly, and those differences drive circulation in the atmosphere and oceans. Warm air rises, cooler air sinks, and large-scale movement begins. That movement helps shape winds, storms, cloud formation, and rainfall.
Land and water also heat at different rates, which affects coastal weather and daily temperature patterns. Thermal energy does not just warm the planet. It helps organize the movement of air and water across the globe.
Thermal Energy in the Earth
A powerful natural example of thermal energy is geothermal energy. This is heat that flows continuously from the Earth’s interior to the surface. It has been doing so for about 4.5 billion years, and it is continually replenished by radioactive decay inside the planet.
Geothermal energy can be used for heating, cooling, and electricity generation. Geothermal heat pumps take advantage of the relatively stable temperature underground, while direct-use systems can provide heat for buildings and industrial processes.
This matters because geothermal energy is a natural, steady source of thermal energy that does not depend on sunshine or wind in the same way many other renewable resources do.
Thermal Energy in Industry
Industry uses a huge amount of thermal energy. It is needed for drying, sterilization, smelting, cement production, food processing, chemical production, and many other processes that transform raw materials into useful products.
One important trend is the use of solar-thermal power. This approach concentrates sunlight to produce heat, which can then be used directly in industrial processes or stored for later use. Some systems can reach temperatures from below 400°C to over 1000°C, depending on the technology, and certain industrial process heat applications can be served efficiently by this route.
Thermal energy also plays a central role in combined heat and power systems and in heat recovery, where waste heat is reused instead of being thrown away. That makes industrial systems more efficient and can reduce fuel use.
Thermal Energy Storage
One of the smartest ways to use thermal energy is to store it. Thermal energy storage means keeping heat for later use, whether for heating, cooling, power generation, or industrial work. It can improve reliability, shift energy use to more convenient times, and make renewable systems more practical.
Storage systems are often grouped into three broad types. Sensible heat storage stores energy by raising the temperature of a material. Latent heat storage stores energy during a phase change. Thermochemical storage stores thermal energy in chemical bonds.
Table: Thermal Energy Storage Types
| Storage type | How it stores energy | Typical example | Main advantage |
|---|---|---|---|
| Sensible heat storage | Raises the temperature of a material | Hot water tanks, chilled water tanks | Simple and widely used |
| Latent heat storage | Stores energy during melting or freezing | Phase change materials, ice storage | High storage density with smaller temperature swings |
| Thermochemical storage | Stores energy in reversible chemical reactions | Salt hydrate systems and other reactive materials | High energy density and low self-discharge losses |
A simple example of sensible storage is a hot water tank. Water can be heated during low demand and used later when heat is needed. This is common in buildings and combined heat and power systems.
A simple example of latent storage is a phase change material. These materials absorb or release large amounts of thermal energy while changing state, which helps regulate temperature efficiently.
A simple example of thermochemical storage is a salt-based system that stores energy in chemical bonds and releases it later when the reaction is reversed. This approach can provide high energy density and good long-term storage performance.
Thermal Energy and Human Comfort
Our bodies are always exchanging thermal energy with the environment. We lose heat to cool air, gain heat from sunlight, and rely on sweating, clothing, airflow, and shelter to stay in balance. That is why thermal comfort is such a big part of health and daily life.
A warm room can still feel unpleasant if the humidity is too high or if the air does not move well. A cooler room can feel comfortable if it is well ventilated. That shows thermal comfort is not only about temperature. It is also about how thermal energy moves around us.
Everyday Examples of Thermal Energy
Here are some familiar situations where thermal energy is easy to notice:
- Cooking food uses thermal energy to change texture, soften ingredients, and raise the temperature.
- A refrigerator removes thermal energy from the inside compartment and sends it out through the system.
- A radiator or heater transfers thermal energy to a room through air movement and surface heating.
- A blacktop road in the sun absorbs thermal energy and warms faster than many lighter surfaces.
- A blanket slows heat loss by reducing the movement of thermal energy away from the body.
Common Myths About Thermal Energy
One myth is that heat and temperature are the same thing. They are not. Temperature measures a state, while heat is energy in transfer.
Another myth is that cold is a thing that moves in. In physics, what usually happens is that thermal energy moves out of a warmer object into a cooler one. The cooler object does not “send cold” into the warmer one.
A third myth is that only hot things have thermal energy. In reality, every material object above absolute zero has some thermal energy because its particles are in motion.
Why Thermal Energy Is So Important
Thermal energy sits at the center of basic science and modern technology. It explains how the atmosphere works, how buildings stay comfortable, how engines lose energy, how food cooks, how Earth stores heat, and how renewable systems can be designed for storage and flexibility.
It also connects directly to energy efficiency. The better we understand thermal energy, the better we can design insulation, heat exchangers, refrigeration systems, power plants, and storage technologies. In a world that depends heavily on energy, that understanding is not optional. It is essential.
A Simple Summary You Can Remember
Thermal energy is the energy tied to the motion of particles inside matter. Heat is thermal energy in motion from a warmer place to a cooler one. Temperature tells us how energetic the particles are on average. And thermal energy storage is the practical art of keeping heat for later use.
If you remember just that, you already understand the heart of the topic. From a spoon in a cup of coffee to a geothermal system under the ground, thermal energy is everywhere, quietly shaping the way the world works.
Final Thoughts
Thermal energy is not a niche scientific idea. It is one of the most practical and visible forms of energy in everyday life. It helps explain comfort, cooking, climate, industry, and technology. It also gives us a way to think clearly about heat, temperature, insulation, phase change, and energy storage without confusion.
Article References and Sources
- Beeindia: Thermal Energy Basics and Heat Transfer Concepts
- NIST: Kelvin and Thermodynamic Temperature (Measurement of Temperature)
- Energy: Principles of Heating and Cooling (Heat Transfer Explained)
- NOAA: Heat Transfer Educational Material (Conduction, Convection, Radiation)
- DOE: Energy Transfer and Thermodynamics Concepts (Educational Resource)
- DOE: Thermal Energy Storage Technology Strategy Assessment
- DOE: Insulation and Energy Efficiency in Buildings
- NIST: Thermophysical Properties and Specific Heat Data
- NOAA: Radiative Heating of Land and Water (Climate and Thermal Energy)
- DOE: Geothermal Energy FAQs (Earth’s Internal Heat)
- DOE: Geothermal Energy Basics
- DOE: Solar Thermal Power and Industrial Process Heat
- DOE: Combined Heat and Power Systems Overview
- DOE: Thermal Energy Storage Systems Overview
- DOE: Thermal Energy Storage Fact Sheet
- DOE: Phase Change Materials for Thermal Energy Storage
- DOE: Thermochemical Energy Storage Materials Development
- BIPM: CGPM Resolution on the Joule (Unit of Energy)
Also, Read these Articles in Detail
- Physics and Its Fundamentals With Good Explanations
- Matter, Motion, and Energy: The Core Ideas of Physics
- What Is Matter? The Physical Substance of the Universe
- What Is Motion? A Guide to Motion in Physics and Daily Life
- What Is Energy? The Invisible Power Behind Everyday Life
- Kinetic Energy Explained in Simple Language
- Potential Energy: Definition, Types, Formula, and Examples
Frequently Asked Questions
FAQ 1: What is thermal energy in simple words?
Thermal energy is the energy inside a material that comes from the motion of its tiny particles, like atoms and molecules. These particles are always moving in some way. In a solid, they mostly vibrate in place. In a liquid, they move around more freely. In a gas, they move much faster and spread out. The more active this particle motion is, the more thermal energy the substance has.
This is why a hot object feels hot. Its particles are moving more quickly than the particles in a colder object. That movement is not something we can see directly, but we can feel its effects every day. When you hold a warm cup of tea, sit in sunlight, or touch a heated pan, you are dealing with thermal energy in action. It is one of the most common forms of energy in life, and it shows up everywhere.
People often think thermal energy is the same as heat, but that is not quite right. Thermal energy is the energy inside the object. Heat is the energy that moves from one object to another because of a temperature difference. So, thermal energy is the stored energy, while heat is the transfer of that energy.
You can also think of thermal energy as one reason materials change temperature, melt, boil, or cool down. When energy enters a substance, the particles move faster. When energy leaves, they slow down. That simple idea helps explain a huge part of physics, chemistry, weather, cooking, and engineering. And that is why thermal energy matters so much in everyday life.
FAQ 2: How is thermal energy different from heat and temperature?
This is one of the most important questions in science because these three words are often mixed up. They are related, but they do not mean the same thing.
Temperature is a measure of how fast the particles in a substance are moving on average. If the particles are moving faster, the temperature is usually higher. If they are moving more slowly, the temperature is usually lower. Temperature tells us how hot or cold something is, but it does not tell us everything about the total energy in the object.
Heat is energy that moves from a warmer object to a cooler one because of a temperature difference. Heat is not something an object “has” in the same way it has mass. Heat is a process. It describes energy in motion. For example, when a hot pan warms a cold spoon, thermal energy is moving from the pan to the spoon. That movement is heat transfer.
Thermal energy is the energy connected to the motion of particles inside the material. A big object and a small object can have the same temperature but very different amounts of thermal energy. A bathtub of warm water may contain much more thermal energy than a tiny drop of boiling water, even though the drop is hotter.
That difference matters in real life. A large bowl of soup can stay warm longer than a tiny cup because it has more total thermal energy. A metal spoon in hot soup may heat up faster than the soup changes temperature. A sunny sidewalk can feel much hotter than the surrounding air because the surface absorbs thermal energy directly.
So the simplest way to remember it is this:
- Temperature tells you how hot something is.
- Heat is energy moving from one place to another.
- Thermal energy is the energy inside the matter itself.
FAQ 3: How does thermal energy move from one object to another?
Thermal energy moves in three main ways, and each one works differently. These are conduction, convection, and radiation. Once you understand these three, a lot of everyday science becomes much easier to follow.
Conduction happens when thermal energy moves through direct contact. If one part of an object gets hot, the energy can pass to nearby particles by collision and vibration. This is why a metal spoon in a hot drink becomes warm. The heat does not jump through the spoon magically. It moves particle by particle through the material. Metals are especially good at conducting thermal energy, which is why they are used in cookware and heat exchangers.
Convection happens in fluids, which means liquids and gases. When a fluid is heated, it expands and becomes less dense, so it rises. Cooler, denser fluid moves down to replace it. This creates a cycle called a convection current. You can see this in boiling water, in warm air rising in a room, and even in large-scale weather systems.
Radiation is different because it does not need direct contact or even a material medium. Thermal energy moves as electromagnetic waves, especially infrared radiation. This is how the Sun warms the Earth. Space is nearly empty, so conduction and convection cannot happen there. But radiation can still travel across space and bring energy to our planet.
All three forms of transfer happen around us all the time. A hot stove uses conduction and radiation. Boiling water uses convection. Sunlight warms the ground by radiation. In real life, these modes often work together, not separately. That is why thermal energy is so central to both nature and technology.
FAQ 4: Why do some materials heat up faster than others?
Different materials respond to thermal energy in different ways. Some heat up quickly. Some stay warm for a long time. Some let heat pass through easily. Others slow heat down. This depends on the material’s structure, its specific heat capacity, and how easily energy moves through it.
Specific heat capacity is the amount of thermal energy needed to raise the temperature of a substance by a certain amount. Materials with a high specific heat need more energy to change temperature. Materials with a low specific heat change temperature more quickly. This is one reason why water behaves very differently from metal under the same conditions.
Metals usually heat up and cool down quickly because they conduct thermal energy well. That is why a metal spoon left in a hot pan can become too hot to touch in a short time. Water, on the other hand, can absorb a lot of thermal energy before its temperature changes very much. That makes it useful in heating systems, cooling systems, and climate regulation.
The color and texture of a material can also matter. Dark surfaces often absorb more radiation than light surfaces, which is why blacktop roads get hotter in the sun than many lighter-colored surfaces. Smooth shiny surfaces may reflect more radiation, while rough or dark surfaces may absorb more.
In practical terms, this is why people use different materials for different jobs. Copper and aluminum are useful in cookware because they transfer heat efficiently. Foam, fiberglass, and trapped air are useful in insulation because they slow heat flow. And water is useful in thermal storage because it can hold a lot of energy without changing temperature too fast.
FAQ 5: What happens when thermal energy is added to a substance?
When thermal energy is added to a substance, several things can happen. The most obvious result is that its temperature may rise. But that does not always happen right away, and this is where the science gets more interesting.
If the added energy goes into speeding up the particles, then the temperature increases. That is what happens when a cup of water slowly warms on the stove. The particles inside gain more motion, and the thermometer rises. But sometimes the energy does something else. It can go into changing the state of matter instead of raising the temperature.
For example, when ice melts, thermal energy is absorbed, but the temperature may stay near the melting point until the change is complete. The energy is being used to break the forces holding the solid structure together. The same idea happens during boiling. Water can absorb a lot of energy while turning into steam without the temperature rising much beyond the boiling point.
This is why thermal energy is so important in cooking, climate, and engineering. It can change the state of a material, not just its temperature. That is also why thermal energy storage systems are so useful. They can store energy in a form that is later released when needed.
In short, when thermal energy is added, it may:
- raise the temperature
- cause melting
- cause boiling
- make materials expand
- change how a material behaves physically
That is a lot of power from something as ordinary as heat.
FAQ 6: How does thermal energy affect everyday life?
Thermal energy is part of daily life in ways people often do not notice at first. It affects how we cook, how we dress, how we travel, how we stay comfortable, and how buildings are designed. It also affects weather, food storage, and energy use.
In the kitchen, thermal energy cooks food by changing its structure and texture. Eggs firm up, bread browns, vegetables soften, and water turns into steam. The whole process depends on controlling how much thermal energy is transferred and how quickly it moves.
In homes, thermal energy helps determine whether a room feels comfortable or too hot or too cold. Insulation, windows, roofing, and even curtains all affect thermal energy flow. In winter, we want to keep thermal energy inside. In summer, we want to keep excess thermal energy out.
In clothing, the same principles apply. A thick jacket slows the movement of thermal energy from your body to the cold air outside. A blanket does the same thing while you sleep. This is why warm clothes are not really “making heat” themselves. They are helping your body keep its own thermal energy.
In transportation, thermal energy matters in engines, brakes, batteries, and cooling systems. Cars, trains, airplanes, and even electric vehicles must manage heat well or they become inefficient or unsafe. So thermal energy is not just a science idea. It is part of everyday living from morning to night.
FAQ 7: Why is thermal energy important in weather and climate?
Thermal energy is one of the main forces behind weather and climate. The Sun gives Earth thermal energy, but it does not heat the entire planet evenly. Some areas get more sunlight than others. Land and water also absorb and release thermal energy differently. These differences drive movement in the atmosphere and oceans.
Warm air rises because it becomes less dense. Cooler air moves in to replace it. That movement creates wind and helps form weather systems. When warm, moist air rises and cools, it can form clouds and storms. So the motion of thermal energy is a big part of daily weather patterns.
Oceans also store huge amounts of thermal energy. Water has a high heat capacity, so large bodies of water warm up and cool down more slowly than land. This helps moderate temperatures in coastal regions. It is one reason beaches often feel cooler during the day and warmer at night compared with inland areas.
Climate is also shaped by how thermal energy is trapped or released in the atmosphere. Greenhouse gases affect the movement of infrared radiation, which changes how much thermal energy stays near Earth’s surface. That is why thermal energy is closely connected to climate science, environmental studies, and global warming discussions.
So when people talk about wind, storms, seasons, ocean currents, or climate change, thermal energy is always part of the picture. It is one of the hidden engines of the planet.
FAQ 8: What is thermal energy storage and why is it useful?
Thermal energy storage means storing heat or cold for later use. This is a very practical idea because not all energy needs happen at the same time as energy production. Sometimes energy is available now but needed later. Thermal storage helps solve that problem.
There are three major kinds of thermal storage. Sensible heat storage works by changing the temperature of a material, like heating water in a tank. Latent heat storage works by using a material that changes state, such as melting or freezing. Thermochemical storage uses reversible chemical reactions to store thermal energy in a more advanced way.
This matters in homes, factories, solar systems, and large buildings. A hot water tank can store heat for later use. Ice storage can help cool buildings during the day after being produced at night. Some industrial systems store thermal energy from solar collectors and use it when sunlight is not available.
Thermal storage is useful because it improves efficiency, reduces wasted energy, and helps match supply with demand. It can also support renewable energy systems, since sunlight and heat can be stored instead of used immediately. That makes energy systems more flexible and reliable.
A simple way to think about it is this. Thermal energy storage is like saving warmth for later. Instead of letting heat disappear, we hold on to it until it is needed. That is a very smart idea, and it is becoming more important every year.
FAQ 9: What are some real-life examples of thermal energy storage and thermal control?
There are many real-life examples of thermal energy storage and thermal control around us, even if we do not always notice them.
A thermos flask keeps drinks hot or cold by slowing thermal energy transfer. It reduces conduction, convection, and radiation so the drink stays closer to its original temperature. A refrigerator removes thermal energy from the inside compartment and releases it outside using a cooling cycle. A blanket traps air and slows heat loss from the body. A cooling tower in industry releases unwanted thermal energy from machinery and systems.
In buildings, insulation helps control thermal energy by reducing heat flow through walls, ceilings, and roofs. Double-pane windows, reflective roofing, and shaded windows all help manage thermal energy more effectively. These are not just comfort features. They are energy-saving tools.
In power systems, some solar plants use thermal storage to capture heat during the day and release it later. In food systems, cold storage and freezers preserve thermal conditions that slow spoilage. In medicine, temperature-controlled storage protects vaccines and sensitive materials.
These examples show that thermal energy is not only a science concept. It is part of design, engineering, safety, and everyday problem-solving. Whenever we store, redirect, or block heat, we are working with thermal energy.
FAQ 10: Why should students and readers understand thermal energy well?
Understanding thermal energy gives you a stronger grasp of science, but it also helps in many practical situations. Once you know how thermal energy works, you can better understand cooking, weather, insulation, engines, cooling, heating, and energy conservation. It turns a lot of confusing things into clear ideas.
For students, thermal energy is one of the building blocks of physics and thermodynamics. It connects to particle motion, energy transfer, phase changes, and material properties. These are topics that show up again and again in science classes. If you understand thermal energy early, the rest becomes easier to follow.
For everyday life, the knowledge is just as useful. It helps you choose better clothes in hot or cold weather, understand why some materials feel warmer than others, manage electricity use at home, and even make smarter decisions about cooking and insulation. It also helps you understand the world more calmly and clearly.
Thermal energy is everywhere. It is in the Sun, in the Earth, in your kitchen, in your body, in weather systems, and in modern technology. Once you start noticing it, the world feels a little more connected. Things that seemed separate begin to make sense together. And that is the real value of learning about thermal energy. It gives you a simple way to understand a very large part of life.