Chemical energy is one of the most important forms of energy in everyday life. It is the energy stored in the bonds between atoms and molecules, and it is released or absorbed during chemical reactions. You use it every day, often without thinking about it. It powers your body, runs cars, lights stoves, charges phones, and helps industries produce countless materials that people depend on.
When people hear the word energy, they often think of electricity, fuel, or motion. But chemical energy sits behind many of those things. A piece of wood in a fire, a battery in a flashlight, a banana eaten before a workout, and gasoline in a car all involve chemical potential energy. That means the energy is stored in chemical substances and can be released when those substances change.
This article gives a clear, detailed, and practical explanation of chemical energy. It covers what it is, how it works, where it appears in nature, how humans use it, why it matters, and how it connects to science, technology, and daily life. The language stays simple, but the detail goes deep enough to give a real understanding of the topic.
Table of Contents
What Is Chemical Energy?
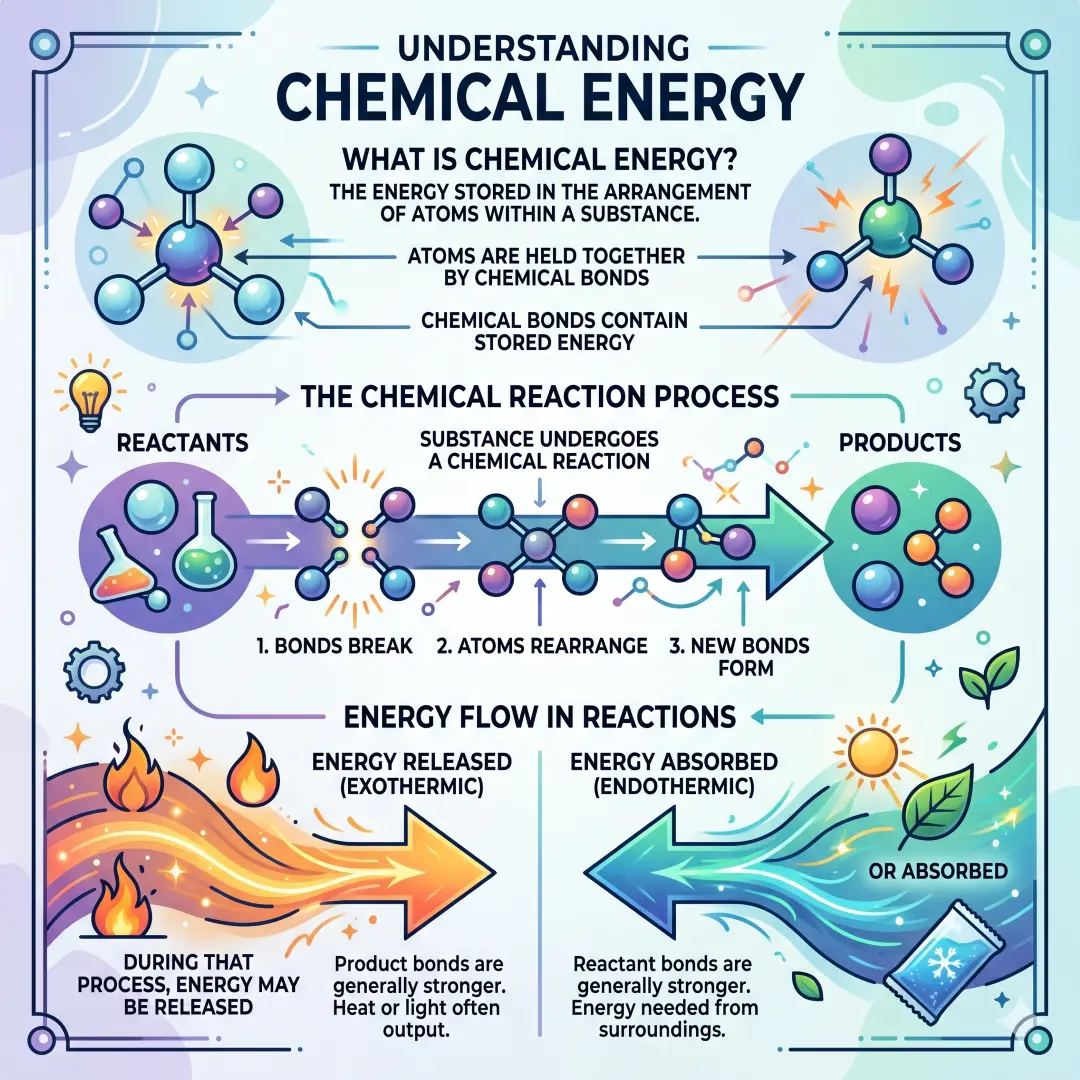
Chemical energy is the energy stored in the arrangement of atoms within a substance. Atoms are held together by chemical bonds, and those bonds contain stored energy. When a substance undergoes a chemical reaction, the bonds break and new bonds form. During that process, energy may be released or absorbed.
A simple way to think about it is this. Chemical energy is like stored power waiting for the right moment to be used. Sometimes that energy is released quickly, like in combustion. Sometimes it is released slowly, like in digestion. And sometimes it is stored in a stable form for a long time, like in a battery that is not yet being used.
Chemical energy is a form of potential energy because it depends on position and arrangement, not on motion alone. It is different from kinetic energy, which is the energy of movement. Chemical energy becomes useful when it changes into other forms such as heat, light, sound, electrical energy, or mechanical energy.
The Science Behind Chemical Energy
To understand chemical energy properly, it helps to know a little about atoms and bonds.
Everything around you is made of atoms. These atoms join together to form molecules and compounds. They do not join randomly. They form bonds because that arrangement often gives them a more stable structure.
The energy stored in those bonds comes from the forces between charged particles in the atom. When atoms bind together, energy is involved. Some bonds are strong and stable. Others are easier to break. When a reaction happens, the difference between the energy in the old bonds and the energy in the new bonds determines whether energy is released or absorbed.
In simple terms:
- If breaking old bonds takes less energy than forming new bonds gives back, energy is released.
- If breaking old bonds takes more energy than new bonds release, energy is absorbed.
That is why some reactions feel hot, while others feel cold.
Chemical Energy as Stored Potential Energy
Chemical energy is usually described as stored potential energy. This means the energy is not being used yet, but it can be released when conditions change.
Think of it like this:
- A battery stores chemical energy until a circuit is completed.
- Food stores chemical energy until your body breaks it down.
- Gasoline stores chemical energy until it burns in an engine.
- Wood stores chemical energy until it catches fire.
The energy is there because of the structure of the substance. Once the substance reacts, that energy can be transformed into other forms.
Where Chemical Energy Is Found in Daily Life
Chemical energy is everywhere. In fact, it would be hard to find a normal day that does not involve it.
Examples from everyday life
- Food gives your body energy.
- Batteries power remotes, phones, toys, and flashlights.
- Fuel powers cars, buses, airplanes, and generators.
- Matches release energy when struck.
- Natural gas burns for cooking and heating.
- Cleaning products often depend on chemical reactions.
- Medicines work through chemical processes in the body.
You may not see chemical energy directly, but you see its effects all the time. A cup of tea heated on a stove, a running motorcycle, a lit candle, or a person jogging after breakfast all involve chemical energy being transformed.
How Chemical Energy Changes Form
One of the most interesting things about chemical energy is that it does not disappear. It changes form.
For example:
- In a candle, chemical energy in wax becomes heat and light.
- In your body, chemical energy in food becomes movement, body heat, and stored energy.
- In a battery, chemical energy becomes electrical energy.
- In a car engine, chemical energy in fuel becomes mechanical energy and heat.
This follows a basic scientific idea known as the law of conservation of energy. Energy cannot be created or destroyed. It only changes form.
Chemical Reactions and Energy
Chemical energy becomes useful through chemical reactions. These reactions can either release energy or absorb it.
Exothermic reactions
An exothermic reaction releases energy, usually as heat. These reactions often feel warm or hot.
Examples include:
- Burning wood
- Combustion of gasoline
- Respiration in the body
- Neutralization reactions in some cases
Endothermic reactions
An endothermic reaction absorbs energy from the surroundings. These reactions often feel cool.
Examples include:
- Some forms of photosynthesis
- Certain laboratory reactions
- Instant cold packs used for injuries
Both types matter. They help explain why some reactions produce heat and others need heat to happen.
Chemical Energy in Food
Food is one of the best examples of chemical energy in action. The body takes in food, breaks it down, and extracts energy from it. That energy is then used for everything from thinking and breathing to walking and growing.
Carbohydrates, fats, and proteins all contain chemical energy, but they store it in different ways.
Main energy-rich nutrients in food
- Carbohydrates provide quick energy
- Fats provide concentrated, long-lasting energy
- Proteins mainly help build and repair tissues, but they can also be used for energy if needed
The body uses digestion and cellular respiration to convert food energy into ATP, which is the main energy currency in cells.
Simple food examples
- Rice, bread, and potatoes release energy relatively quickly.
- Nuts, oils, and butter store a lot of energy in a small amount of food.
- Fruits provide energy with vitamins, fiber, and natural sugars.
- Meat, beans, and eggs give both energy and building material for the body.
Without chemical energy in food, the body could not move, think, or stay warm.
Chemical Energy in the Human Body
The human body depends on chemical energy every second of the day. Even when you are sleeping, your body still needs energy to keep breathing, maintain temperature, and repair cells.
Food is broken down into smaller molecules like glucose. Then, inside cells, that glucose is used in cellular respiration to release energy. That energy is stored in ATP, which cells use for almost everything they do.
What chemical energy in the body powers
- Muscle movement
- Brain activity
- Heart function
- Digestion
- Temperature control
- Cell repair and growth
This is why eating well matters so much. Your body is constantly converting chemical energy into work.
Chemical Energy in Batteries
Batteries are a common and useful example of chemical energy. They store energy chemically and release it as electrical energy when connected to a circuit.
Inside a battery, chemical reactions happen between electrodes and an electrolyte. These reactions create a flow of electrons, which is electricity.
Everyday battery-powered items
- Phones
- Remote controls
- Laptops
- Watches
- Flashlights
- Cameras
- Hearing aids
Batteries are so useful because they are portable, reliable, and often rechargeable. In rechargeable batteries, the chemical reactions can be reversed when the battery is charged again.
Chemical Energy in Fuels
Fuels are substances that store large amounts of chemical energy. When they burn, that energy is released and used for heat, electricity, or motion.
Common fuels
- Gasoline
- Diesel
- Natural gas
- Coal
- Wood
- Propane
- Hydrogen in certain energy systems
Fuels are important because they power transportation, industry, and home heating. But they also create environmental challenges, especially when they release carbon dioxide and other pollutants.
Table 1: Common Sources of Chemical Energy and Their Uses
| Source | Where It Is Found | Main Use | Energy Released As | Everyday Example |
|---|---|---|---|---|
| Food | Plants, animals, packaged meals | Powers the human body | ATP, heat, movement | Eating breakfast before school or work |
| Gasoline | Fuel stations, vehicles | Transport | Motion, heat | A car driving on the road |
| Diesel | Trucks, buses, generators | Heavy transport and machinery | Motion, heat | A bus engine running |
| Natural gas | Gas lines, stoves, heaters | Cooking and heating | Heat | Cooking dinner on a stove |
| Wood | Trees, firewood | Heating, cooking, burning | Heat, light | A campfire |
| Batteries | Electronics, devices | Power supply | Electrical energy | A flashlight turning on |
| Coal | Power plants, industrial use | Electricity generation | Heat, then electricity | Large scale power production |
Chemical Energy in Photosynthesis
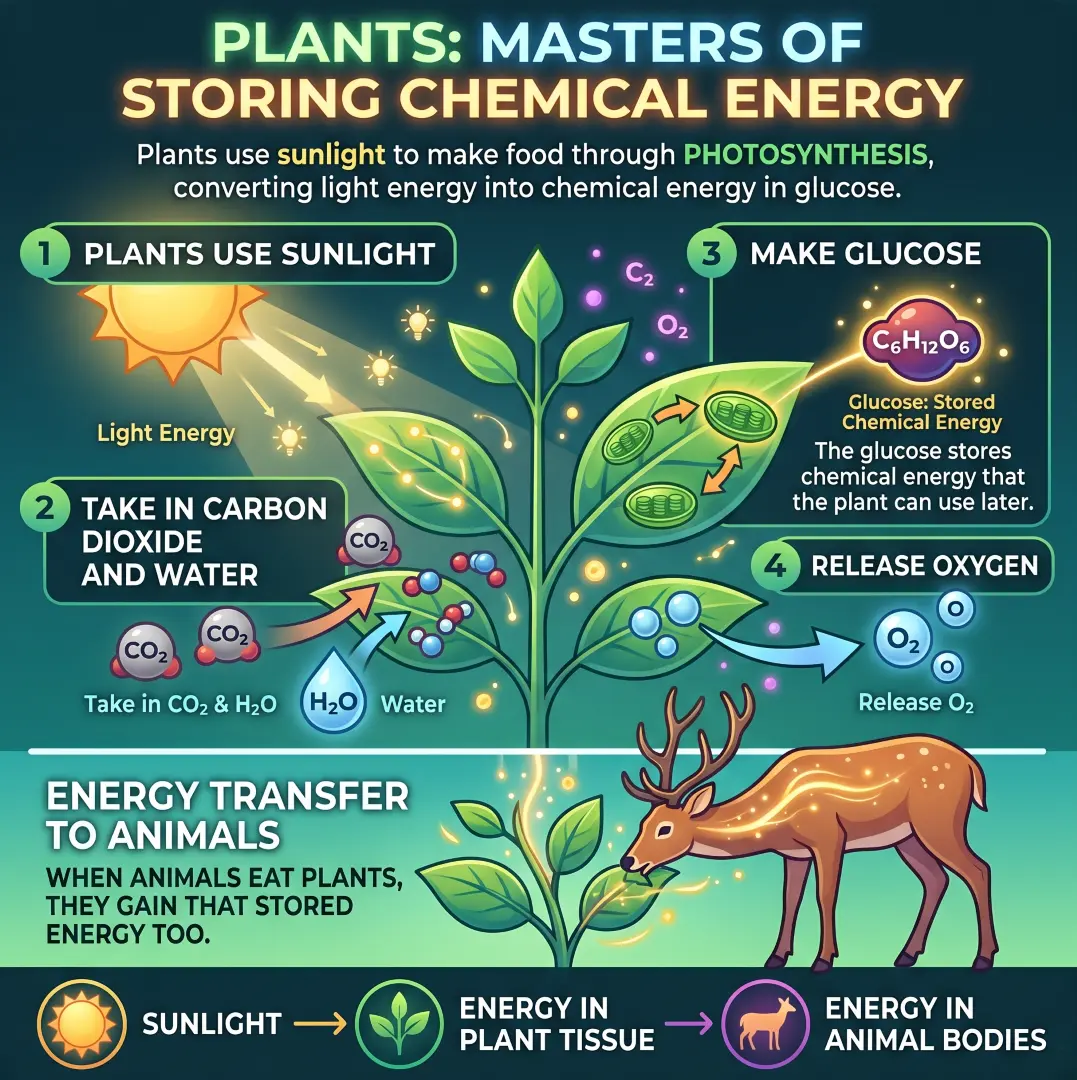
Plants are amazing at storing chemical energy. They take sunlight and use it to make food through photosynthesis. In this process, plants convert light energy into chemical energy stored in glucose.
The basic idea is simple:
- Plants use sunlight
- They take in carbon dioxide and water
- They make glucose
- They release oxygen
The glucose they produce stores chemical energy that the plant can use later. When animals eat plants, they gain that stored energy too. That means sunlight eventually becomes energy in plant tissue, and then energy in animal bodies.
This is one of the most important energy pathways on Earth.
Table 2: Comparison of Chemical Energy in Different Systems
| System | Energy Input | Chemical Energy Stored In | Energy Output | Key Process |
|---|---|---|---|---|
| Plant | Sunlight | Glucose | Food energy, growth | Photosynthesis |
| Human body | Food | ATP | Movement, heat, cell activity | Cellular respiration |
| Battery | Chemical substances inside battery | Reactants at electrodes | Electricity | Electrochemical reaction |
| Car engine | Fuel | Gasoline or diesel | Motion, heat | Combustion |
| Candle | Wax and oxygen | Wax molecules | Light, heat | Burning |
| Power plant | Coal, gas, or other fuels | Fuel compounds | Electricity | Burning and conversion |
Chemical Energy and Combustion
Combustion is a fast chemical reaction between a fuel and oxygen that releases energy. It is one of the most common ways humans use chemical energy.
When something burns, its stored energy changes mostly into heat and light. That is why a burning candle glows, a fire warms a room, and a gas burner heats a pan.
Examples of combustion
- Wood burning in a fireplace
- Gasoline burning in an engine
- Natural gas burning on a stove
- Candle wax burning in air
Combustion is useful, but it must be controlled. Uncontrolled combustion can cause fires, smoke damage, and air pollution.
Chemical Energy and Respiration
Respiration is the process by which living things release energy from food. In humans and many other organisms, this process happens in cells and produces ATP.
Why respiration matters
- It gives energy for movement
- It supports body temperature
- It helps cells repair damage
- It keeps vital organs working
Without respiration, life as we know it would not continue. It is one of the clearest examples of chemical energy being transformed into usable energy.
Chemical Energy in Industry
Industry depends heavily on chemical energy. Factories, machines, and large systems use fuels and chemical reactions to produce goods, power tools, and generate electricity.
Industrial uses include
- Steel production
- Cement manufacturing
- Plastic production
- Chemical manufacturing
- Power generation
- Food processing
In many industries, chemical energy is used because it is dense and easy to store. A small amount of fuel can produce a large amount of heat or power.
Table 3: Chemical Energy vs Other Major Energy Types
| Energy Type | What It Is | Main Example | How It Moves or Works |
|---|---|---|---|
| Chemical energy | Energy stored in chemical bonds | Food, fuel, battery | Released through chemical reactions |
| Kinetic energy | Energy of motion | Moving car, flowing water | Depends on speed and mass |
| Thermal energy | Energy from particle motion in matter | Hot stove, boiling water | Related to temperature |
| Electrical energy | Energy from moving charges | Power lines, batteries | Travels through circuits |
| Mechanical energy | Energy of moving or positioned objects | Windmill, lifted weight | Combines motion and position |
| Radiant energy | Energy carried by light | Sunlight, lamp light | Travels as electromagnetic waves |
| Nuclear energy | Energy stored in atomic nuclei | Sun, nuclear power plants | Released by nuclear changes |
This comparison shows that chemical energy is only one part of the larger energy picture, but it is one of the most practical and familiar.
Why Chemical Energy Matters So Much
Chemical energy matters because it supports life, movement, industry, and technology. Without it, daily life would look very different.
Reasons chemical energy is important
- It powers the human body
- It makes food useful
- It fuels transportation
- It stores power in batteries
- It supports industry
- It helps generate electricity
- It plays a role in medicine and biology
It is hard to overstate how central it is. A modern city depends on countless chemical energy systems running at once.
Chemical Energy in Transportation
Transportation relies heavily on chemical energy, especially in vehicles that burn fuel.
Examples
- Cars use gasoline or diesel
- Airplanes use jet fuel
- Ships often use marine fuel
- Some buses use compressed natural gas
- Hybrid vehicles use both fuel and electric batteries
- Electric vehicles store energy in batteries that still depend on chemistry
The fuel is burned or used in a chemical reaction, and that energy becomes motion. This is why transportation is one of the biggest areas where chemical energy is consumed.
Chemical Energy and the Environment
Chemical energy is useful, but it also affects the environment in important ways. When fuels burn, they release not only energy but also gases and particles.
Environmental concerns
- Carbon dioxide contributes to climate change
- Smog can form from vehicle emissions
- Air pollution can affect human health
- Mining and drilling can damage ecosystems
- Waste from batteries can be harmful if not managed properly
At the same time, chemical energy is part of the solution too. Better batteries, cleaner fuels, and more efficient reactions can reduce pollution.
Cleaner approaches include
- More efficient engines
- Rechargeable batteries
- Biofuels
- Hydrogen technologies
- Improved recycling systems
- Better energy storage solutions
The goal is not to eliminate chemical energy. That would be impossible and unnecessary. The goal is to use it more wisely.
Chemical Energy in Renewable and Low-Carbon Systems
Many people think of chemical energy only in terms of fuels, but it also plays a role in newer energy systems.
Examples
- Hydrogen fuel cells convert chemical energy into electricity
- Batteries store energy for solar and wind systems
- Biofuels come from plant-based sources
- Synthetic fuels are being developed for lower emissions
These systems matter because they can help reduce dependence on older, more polluting fuels. They also show that chemical energy is not outdated. It is part of the future too.
Everyday Examples That Make Chemical Energy Easy to Understand
Sometimes the best way to understand a topic is to see it in real life.
Simple real-world examples
- A snack before exercise gives your body fuel.
- A battery-powered toy works because chemical energy becomes electricity.
- A campfire releases heat from stored chemical energy in wood.
- A car engine turns fuel into motion.
- A plant stores energy from sunlight in sugar.
- A hand warmer uses a chemical reaction to make heat.
These examples show that chemical energy is not some distant scientific idea. It is part of ordinary life.
Misunderstandings About Chemical Energy
People often confuse chemical energy with other ideas. Clearing that up helps a lot.
Common misunderstandings
- Chemical energy is not only dangerous. It is useful and necessary.
- All chemical reactions do not release energy. Some absorb it.
- Energy does not vanish. It changes form.
- Batteries do not create energy. They convert stored chemical energy into electricity.
- Food does not give instant power by itself. Your body must break it down first.
These ideas are small, but they matter. They help people understand science more accurately.
Table 4: Chemical Energy in Everyday Objects
| Object | Stored Chemical Energy Source | What Happens When Used | Final Energy Form |
|---|---|---|---|
| Flashlight battery | Chemicals inside battery | Circuit allows electron flow | Electrical energy, light |
| Apple | Natural sugars and nutrients | Digestion and respiration | Body energy, heat |
| Candle | Wax | Wax burns in oxygen | Heat, light |
| Matchstick | Chemicals on match head | Ignition from friction | Heat, flame |
| Gas stove | Natural gas | Fuel burns | Heat for cooking |
| Backpack snack bar | Sugars, fats, proteins | Digestion | ATP for body use |
| Gasoline tank | Hydrocarbons | Engine combustion | Motion, heat |
Chemical Energy and Safety
Because chemical energy can be released so easily in some situations, safety matters.
Safety examples
- Keep fuels away from flames
- Store batteries properly
- Do not mix unknown chemicals
- Use matches and lighters carefully
- Follow instructions on household cleaners
- Dispose of batteries properly
A safe understanding of chemical energy is as important as a scientific one. Many accidents happen when people forget that ordinary-looking materials can store a lot of energy.
How Scientists Measure Chemical Energy
Scientists study chemical energy by looking at how much energy is released or absorbed in reactions. They use concepts such as enthalpy, bond energy, and activation energy.
Useful scientific ideas
- Enthalpy helps describe heat change in reactions
- Bond energy refers to the energy needed to break bonds
- Activation energy is the initial energy needed to start a reaction
- Catalysts lower the activation energy and help reactions happen faster
These ideas help chemists design better reactions, better fuels, and better materials.
Chemical Energy in the Future
Chemical energy will continue to matter in the future, even as technology changes. In fact, it may matter even more.
Future possibilities
- Better batteries with longer life
- More efficient fuel cells
- Cleaner biofuels
- Safer chemical storage
- Better energy conversion systems
- Advanced synthetic fuels
As energy needs grow, people will keep looking for ways to store and use chemical energy more cleanly and efficiently.
Why This Topic Is Useful for Students and Readers Everywhere
Understanding chemical energy gives you a stronger grasp of science, health, technology, and the environment. It also helps explain many things that people use every day.
What you gain by understanding chemical energy
- A better understanding of biology
- A clearer view of physics and chemistry
- More awareness of energy use
- A stronger sense of how batteries and fuels work
- A practical view of environmental issues
For students, it builds a strong foundation. For adults, it makes everyday life easier to understand. And for everyone, it reveals just how much of the world runs on stored energy that quietly waits inside materials.
Table 5: Advantages and Limitations of Chemical Energy
| Aspect | Advantages | Limitations |
|---|---|---|
| Storage | Can be stored in compact forms like fuel and batteries | Some forms degrade over time |
| Transport | Easy to move in tanks, batteries, and containers | Some fuels are hazardous |
| Use | Can be converted into heat, motion, or electricity | Not all conversions are efficient |
| Availability | Widely available in food, fuels, and materials | Fossil fuels are finite |
| Practicality | Powers homes, vehicles, and devices | Can create pollution when burned |
| Reusability | Rechargeable batteries can be reused | Many fuels are used once and lost |
Chemical Energy in Simple Words
If all the science terms feel heavy, here is the simplest version:
- Chemical energy is stored energy in substances.
- It is released when substances change in a chemical reaction.
- It powers the body, fuels vehicles, charges batteries, and helps plants grow.
- It is one of the most important forms of energy in the world.
That simple idea connects to food, fire, fuel, and life itself.
Frequently Seen Examples of Chemical Energy in Nature
Nature uses chemical energy in many ways without human help.
Natural examples
- Plants storing energy through photosynthesis
- Animals using food for movement and survival
- Microorganisms breaking down matter and releasing energy
- Volcanic and geothermal systems involving chemical changes in minerals
- Natural decomposition where chemical energy is slowly released
Nature is full of energy transformations. Chemical energy is one of the main ones.
How Chemical Energy Supports Modern Life
Modern life depends on convenience, speed, and power. Chemical energy helps make that possible.
It supports
- Cooking
- Heating
- Travel
- Communication devices
- Medical tools
- Farming equipment
- Factories
- Emergency power systems
A world without chemical energy would not just be inconvenient. It would be almost unrecognizable.
Final Thoughts on Chemical Energy
Chemical energy is one of the most practical and important ideas in science. It is stored in the bonds of atoms and molecules, and it appears everywhere from food and fuel to batteries and living cells. It keeps the body moving, powers machines, helps plants grow, and supports modern technology.
What makes it so interesting is that it is both ordinary and powerful. You can see its effects in a candle flame, a running car, a battery-powered phone, or a person eating lunch before a long day. It is quiet until it is released, but once it is transformed, it shapes the world around us.
Understanding chemical energy gives you a better view of life itself. It explains how nature stores power, how technology uses it, and how science keeps improving the way we live. And the more you notice it, the more you realize that a huge part of the world is built on invisible energy waiting inside matter.
Article References and Sources
- Chemical Energy Definition and Facts (Encyclopaedia Britannica)
- Forms of Energy and Chemical Energy Basics (U.S. Energy Information Administration)
- Chemical Energy and Energy Transformation (BYJU’S)
- Energy Basics and Thermochemistry (Lumen Learning Chemistry)
- Understanding Chemical Energy and ATP (Tara Energy Educational Guide)
- Chemical Energy Scientific Overview (Wikipedia Educational Entry)
- Potential Energy and Chemical Potential Energy (Wikipedia Educational Resource)
- Bond Energy and Chemical Bonds (Wikipedia Educational Resource)
- Chemical Thermodynamics Concepts (Wikipedia Educational Resource)
- Chemical Energy Examples and Reactions (Study.com Educational Resource)
- Chemistry and Energy Concepts (Wikipedia Educational Entry)
- Educational Video on Energy and Chemistry
Also, Read these Articles in Detail
- Physics and Its Fundamentals With Good Explanations
- Matter, Motion, and Energy: The Core Ideas of Physics
- What Is Matter? The Physical Substance of the Universe
- What Is Motion? A Guide to Motion in Physics and Daily Life
- What Is Energy? The Invisible Power Behind Everyday Life
- Kinetic Energy Explained in Simple Language
- Potential Energy: Definition, Types, Formula, and Examples
- Thermal Energy: Heat, Temperature, and Transfer
- Mechanical Energy: Definition, Formula, and Examples
Frequently Asked Questions
FAQ 1: What is chemical energy?
Chemical energy is the energy stored inside the bonds of atoms and molecules. It sits quietly in substances until a chemical reaction happens and the stored energy is released or absorbed. That is why chemical energy is often called a form of potential energy. It is not energy in motion yet, but it has the power to become heat, light, electricity, or movement when conditions change.
You can see chemical energy everywhere in daily life. It is in the food you eat, the fuel that powers vehicles, the batteries in your phone, and the wood that burns in a fire. In each case, the material stores energy in its chemical structure. Once that material reacts, the energy is transformed into another form that people can use.
Chemical energy matters because it supports both life and technology. Your body depends on it to breathe, think, move, and stay warm. Machines depend on it to run engines, generate electricity, and power tools. Plants also use energy, but they store it through photosynthesis, which turns sunlight into chemical energy stored in sugar. That means chemical energy is part of a much larger cycle that connects living things, energy use, and the natural world.
A simple way to think about it is this. Chemical energy is stored power waiting to be released. It is stable in one form, but when a reaction starts, that energy can be converted into something useful. That is why chemical energy is such an important idea in chemistry, biology, physics, and everyday life.
FAQ 2: Where is chemical energy found in everyday life?
Chemical energy is found in many ordinary things people use every day, often without noticing it. One of the most common examples is food. When you eat rice, bread, fruit, meat, or vegetables, your body breaks those foods down and uses the chemical energy inside them to keep you alive and active. That energy helps with everything from walking and talking to thinking and growing.
It is also found in fuels such as gasoline, diesel, natural gas, propane, and wood. These substances store a large amount of energy in their molecules. When they burn, that energy is released as heat, light, and motion. A car engine, for example, uses the chemical energy in fuel to move the vehicle forward. A stove uses the chemical energy in gas to heat food. A fireplace uses the chemical energy in wood to warm a room.
Another everyday source is the battery. Batteries store chemical energy and release it as electrical energy when they are connected to a device. That is why flashlights, remote controls, phones, watches, and toys can work without plugging directly into a wall. The battery’s chemicals react in a controlled way, and that reaction produces electricity.
Chemical energy also appears in smaller, more natural ways. A candle holds chemical energy in wax. A matchstick releases it when struck. A person jogging after breakfast is using the chemical energy from food to power muscles. Even a plant has chemical energy stored in its cells because it uses sunlight to make sugar. So chemical energy is not limited to laboratories or factories. It is part of normal life from morning to night.
FAQ 3: How does chemical energy change into other forms of energy?
Chemical energy changes into other forms of energy through chemical reactions. This is one of the most important ideas in science. When atoms and molecules react, old bonds break and new bonds form. During that process, energy is either released or absorbed. The energy does not disappear. It simply changes form.
In many cases, chemical energy becomes heat and light. That happens in combustion, such as when wood burns in a fire or gasoline burns in a car engine. In a candle, the wax reacts with oxygen and gives off both light and warmth. In your body, chemical energy from food is changed into movement, body heat, and energy stored in ATP, which cells use to work.
Chemical energy can also become electrical energy. That is what happens in a battery. Inside the battery, chemical reactions push electrons through a circuit. Those moving electrons are electricity. This is why a battery can run a phone, a calculator, or a flashlight.
Sometimes chemical energy becomes mechanical energy, which is the energy of motion. For example, a fuel-powered engine converts chemical energy into the movement of wheels. In a power plant, chemical energy in fuel may first become heat, then motion, and then electricity. The exact chain depends on the system, but the basic principle stays the same.
This energy change follows the law of conservation of energy, which says energy cannot be created or destroyed, only transferred or transformed. That law is one of the most important ideas in all of science, and chemical energy is a perfect example of it in action.
FAQ 4: Why is chemical energy important for the human body?
Chemical energy is essential for the human body because it powers every major function that keeps a person alive. The food you eat contains chemical energy stored in carbohydrates, fats, and proteins. Your digestive system breaks that food into smaller molecules, and your cells use those molecules to produce usable energy. That energy is then converted into ATP, the main energy source for cells.
Without chemical energy, your body could not do the simplest things. Your muscles would not move. Your heart would not pump. Your brain would not think. Your lungs would not keep working properly. Even when you are resting, your body still needs energy to repair tissues, keep temperature stable, and support organs. So chemical energy is not just for exercise or heavy work. It is needed all the time.
Different nutrients provide energy in different ways. Carbohydrates are often used for quick energy. Fats provide a dense, long-lasting energy supply. Proteins mainly help build and repair the body, but they can also be used for energy if needed. That is why balanced nutrition matters. The body needs a steady supply of chemical energy, not just one big burst.
A helpful way to picture this is to think of food as fuel for the body. Just as a car needs gasoline or electricity to run, the body needs chemical energy from food to work properly. This is why eating well, staying hydrated, and getting enough nutrients are so important. Chemical energy is at the center of health, growth, and daily life.
FAQ 5: What is the role of chemical energy in food and nutrition?
Chemical energy is one of the main reasons food is useful. Food is not just something that tastes good or fills the stomach. It is a source of stored energy that the body needs to survive and function. When you eat, your body breaks down the food through digestion. Then cells convert that energy into a form they can actually use.
Carbohydrates, fats, and proteins all contain chemical energy, but they do not all work the same way. Carbohydrates such as bread, rice, fruits, and potatoes are often broken down quickly, which makes them a fast source of energy. Fats in nuts, oils, dairy products, and meat store a lot of energy and can keep the body going for longer periods. Proteins in eggs, beans, fish, and meat are especially important for growth and repair, but they can also serve as an energy source when needed.
This is why nutrition is about more than calories on a label. It is about how the body uses stored energy and nutrients. A balanced meal gives the body both immediate and long-term support. For example, a meal with rice, vegetables, and protein gives quick energy, steady fuel, and materials for repair all at once.
Chemical energy in food also explains why eating matters before physical activity. A person going for a long walk, doing sports, or studying for hours needs a reliable energy supply. If the body runs low on chemical energy, tiredness and weakness often follow. So food is really a carefully packaged form of stored energy, and nutrition is the process of managing that energy wisely.
FAQ 6: How do batteries use chemical energy?
Batteries are one of the best everyday examples of chemical energy in action. A battery stores energy in chemical substances inside it. When the battery is connected to a device, a chemical reaction starts. That reaction causes electrons to move through the circuit, and that flow of electrons becomes electrical energy.
Inside a battery, there are usually two main parts called electrodes, along with an electrolyte that helps the reaction happen. The chemicals at the electrodes react in a controlled way. As the reaction continues, one side gives up electrons and the other side receives them. This movement is what creates electric current.
That is why a battery can power so many devices. A remote control, flashlight, phone, laptop, wall clock, and toy all depend on this process. The battery does not create energy from nothing. It simply converts the chemical energy it already stores into electricity that the device can use.
Rechargeable batteries work in a similar way, but with an important difference. When you charge the battery, the chemical reaction is reversed. That restores the stored chemical energy so the battery can be used again. This is what happens in phone batteries, laptop batteries, and electric vehicle batteries.
Batteries are useful because they are portable, compact, and reliable. They show how chemical energy can be stored, controlled, and converted when needed. That makes them one of the most practical inventions in modern life.
FAQ 7: What happens during combustion, and how is it linked to chemical energy?
Combustion is a chemical reaction in which a fuel reacts with oxygen and releases energy. In most cases, the energy appears as heat and light. This is one of the most common ways chemical energy is released in the real world.
When wood burns, its chemical bonds break and rearrange with oxygen from the air. The result is energy in the form of flames, warmth, smoke, and gases. When gasoline burns inside a car engine, the same basic principle applies. The stored chemical energy is released and turned into motion. When natural gas burns on a stove, the energy becomes heat for cooking. A candle works the same way on a smaller scale.
Combustion is powerful because it releases a lot of energy in a short time. That is why it is so useful for heating homes, powering engines, and generating electricity. But it also has risks. If combustion is uncontrolled, it can lead to fire, burns, smoke, and pollution. That is why fuel systems must be handled carefully.
Not all chemical reactions are combustion reactions, but combustion is one of the easiest ways to understand chemical energy. It shows clearly how stored energy in a substance can be released when the substance reacts with oxygen. In simple terms, combustion is chemical energy becoming visible through fire, heat, and motion.
FAQ 8: What is the difference between exothermic and endothermic reactions?
The difference between exothermic and endothermic reactions is how energy moves during the reaction. In an exothermic reaction, energy is released to the surroundings. In an endothermic reaction, energy is absorbed from the surroundings.
An exothermic reaction often feels warm because it gives off heat. Burning wood, lighting a candle, or many forms of fuel combustion are exothermic. The chemical system loses energy, and the environment gains it. That is why a fire can heat a room or a stove can cook food.
An endothermic reaction does the opposite. It takes in energy from the surroundings, so the surroundings can feel cooler. Some chemical reactions in labs are endothermic, and certain cold packs use this principle to reduce swelling in injuries. In these cases, the reaction needs energy to continue, so it pulls that energy from nearby materials or the air.
Both types of reactions matter because they show that chemical energy is not always released in the same way. Sometimes energy comes out. Sometimes energy must go in first. The direction of energy flow tells scientists a lot about how a reaction behaves.
Understanding this difference helps make chemical energy easier to see in daily life. A hot pan on a stove, a cold pack on a sore muscle, and a burning log in a fireplace are all examples of energy moving in different directions during chemical change.
FAQ 9: How does chemical energy support plants and photosynthesis?
Chemical energy is central to photosynthesis, the process plants use to make their own food. Plants take in sunlight, water, and carbon dioxide, then use that light energy to build glucose, a sugar that stores chemical energy. This means plants transform light energy into stored chemical energy.
That stored energy is important for the plant itself. It uses the glucose for growth, repair, and survival. Some of that sugar may be stored for later use, and some is used right away. This is how plants keep living and developing. Leaves, stems, roots, flowers, and fruits all depend on this energy process.
Photosynthesis is also important for animals and humans because we depend on plants directly or indirectly for food. When a person eats a plant or eats an animal that ate plants, they are ultimately getting energy that began with sunlight and was stored as chemical energy. That makes plants a key part of Earth’s energy system.
Photosynthesis also produces oxygen, which living things need for respiration. So plants do more than store energy. They help support life across the planet. In that sense, chemical energy in plants is one of the most important links between sunlight, food, air, and living systems.
FAQ 10: Why does chemical energy matter in modern life and the future?
Chemical energy matters in modern life because it supports almost everything people do. It powers the body through food, runs transportation through fuel, operates electronics through batteries, and helps industries produce goods and services. Without chemical energy, modern life would slow down dramatically.
It also matters because energy use is changing. People want cleaner, safer, and more efficient ways to store and use energy. That is why chemical energy is still highly relevant in the future. New battery designs, fuel cells, biofuels, and cleaner chemical systems are all part of ongoing work to improve how energy is stored and released.
In the future, chemical energy may play an even bigger role in electric vehicles, renewable energy storage, medical devices, and portable technology. Batteries will need to last longer and charge faster. Fuel systems will need to release less pollution. New materials may help store more energy in smaller spaces. All of this depends on chemistry.
Chemical energy also remains important because it is so flexible. It can be stored in food, fuel, and batteries. It can be released as heat, motion, or electricity. And it can be used in both natural systems and human-made systems. That is a rare kind of usefulness.
So the future of energy is not just about one source. It is about using chemical energy better, cleaner, and more wisely. And that makes it one of the most important science topics to understand today.